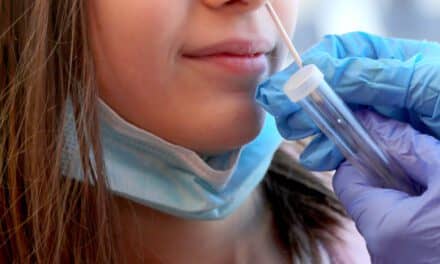
A lateral flow prototype allows for rapid testing at the point of care using capillary blood samples.
VolitionRx Ltd announced the successful detection of nucleosomes in capillary blood from critically ill sepsis patients using a lateral flow prototype. This finger-prick sample test is designed for use at the bedside, in the emergency department, or in a self-test kit, potentially moving testing beyond centralized laboratories.
The study, which is part of the SUMMIT program, tested capillary blood samples from hospital patients in intensive care units. The results demonstrated the feasibility of detecting immune disruptions associated with sepsis simply and rapidly without the need to send a blood sample to a hospital laboratory. Volition previously demonstrated a correlation between whole venous blood samples using the prototype and its established automated Nu.Q nucleosome assay.
“This prototype technology has the exciting potential to strengthen our product portfolio by addressing critical unmet diagnostic needs with an accessible, cost-effective solution enabling rapid, minimally invasive, point-of-care self- testing,” says Gaetan Michel, chief operating officer at Volition, in a release.
Nucleosomes serve as a marker of NETosis and diseases such as sepsis. The company aims to provide a quantitative readout to facilitate clinical decision-making on a low-cost platform.
“The ability to rapidly identify high-risk patients at the point-of-care by quantifying their nucleosome levels using a finger-prick sample and simple lateral flow device could enable quicker clinical decision making and consequently better patient outcomes,” says Cameron Reynolds, CEO at Volition, in a release.
According to the company, recent estimates indicate approximately 166 million cases of sepsis worldwide. All-cause sepsis-related deaths in 2021 represented 31.5% of total global deaths, with the highest burden of mortality in lower-middle-income countries.
“This is a potential game-changer, not only in diseases where time is critical, such as sepsis, but also in significantly expanding potential use cases beyond traditional hospital infrastructure,” says Reynolds in a release.
The project is supported by the Walloon Region and focuses on developing a capillary blood-based lateral flow test for early diagnosis. Volition’s existing Nu.Q nucleosome assay is a chemiluminescent immunoassay that currently runs on the Immunodiagnostic Systems i10 automated analyzer platform. It holds a CE Mark to aid in the detection and evaluation of diseases associated with NETosis.
Photo caption: Nu.Q Lateral Flow Prototype
Photo credit: VolitionRx