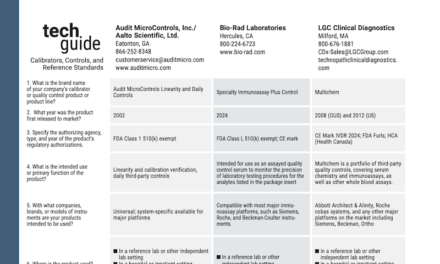
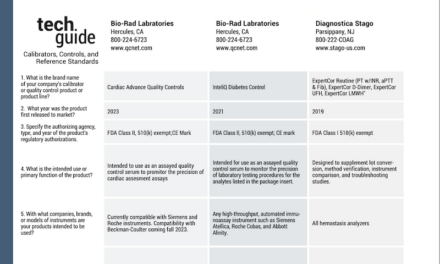
bioMérieux Acquires Accellix for Cell and Gene Therapy Quality Control
bioMérieux has acquired Accellix for about €35 million, adding rapid, automated flow cytometry to its pharmaceutical quality control portfolio for cell and gene therapy manufacturing.