Volition Reports Successful Finger-Prick Detection of Nucleosomes for Sepsis Testing
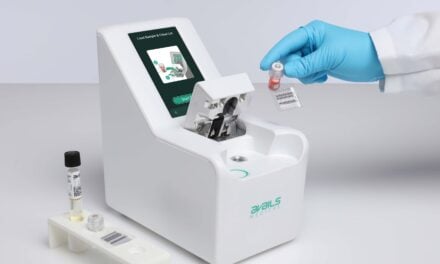
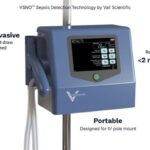
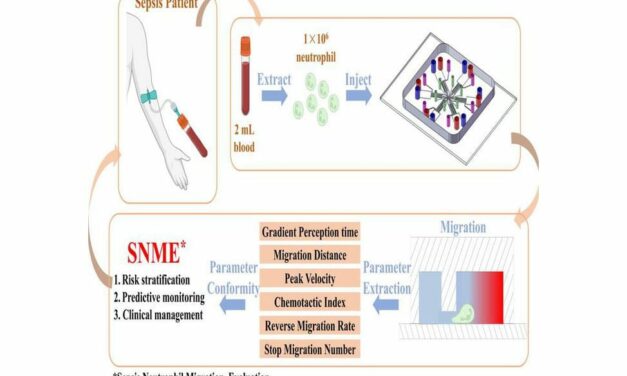
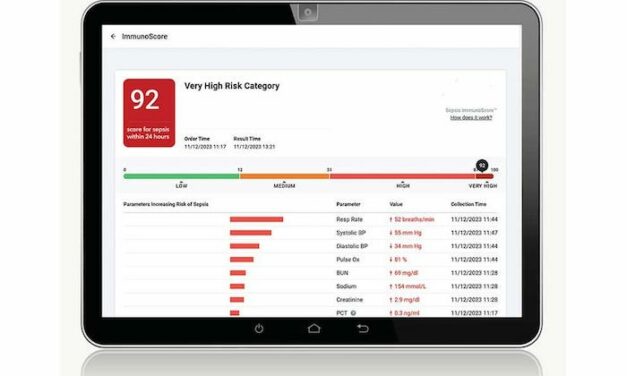
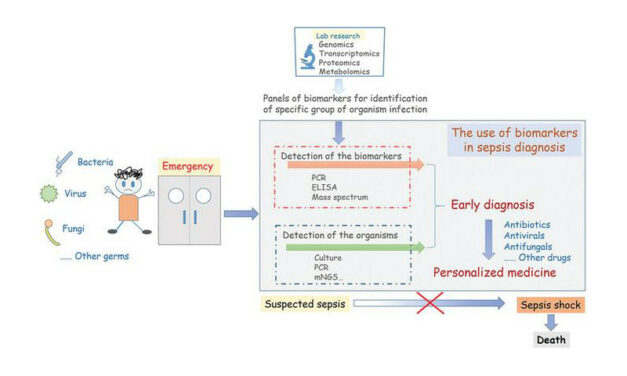
VolitionRx reports its lateral flow prototype can detect nucleosomes in finger-prick blood samples from sepsis patients, enabling rapid, point-of-care testing outside centralized labs, according to company data.