For the first time, WHO is recommending a new class of molecular tests and alternative specimen collection methods to close persistent gaps in global tuberculosis testing.
The World Health Organization (WHO) has issued its first-ever recommendations on near-point-of-care (NPOC) molecular tests, tongue swab specimens, and sputum pooling strategies for the diagnosis of tuberculosis (TB), marking an update to the agency’s consolidated guidelines on TB diagnosis.
The new guidance, which will be formally published as the WHO Consolidated Guidelines on Tuberculosis, Module 3: Diagnosis, 2nd edition, addresses longstanding barriers that have left millions of patients without timely TB diagnosis, including reliance on sputum-based testing, limited availability of laboratory-based platforms at peripheral care sites, and the high costs of existing test technologies.
“These new WHO recommendations mark a major step forward in making TB testing faster and more accessible,” says Tereza Kasaeva, MD, director of WHO’s Department for HIV, TB, Hepatitis & STIs, in a release. “WHO urges countries and partners to work together to roll out these guidelines to close persistent diagnostic gaps and ensure that everyone with TB can be diagnosed early and start life-saving treatment without delay.”
Three New Recommendations at a Glance
The updated guidelines introduce three recommendations for the initial diagnosis of TB:
Near-point-of-care nucleic acid amplification tests (NPOC-NAATs): A new class of molecular tests is now recommended for initial detection of TB — without rifampicin resistance — at peripheral levels of the health system, including peripheral laboratories, primary healthcare centers, and community settings. These tests are designed to operate at lower unit costs than other molecular test and instrument types currently in use.
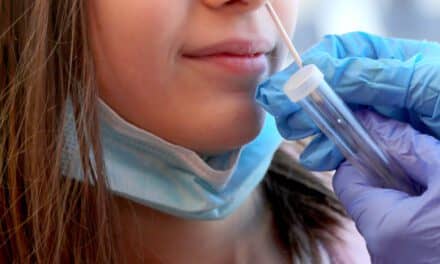
Tongue swabs: WHO now recommends tongue swabs as an alternative specimen type for use with NPOC-NAATs and low-complexity automated NAATs (LC-aNAATs) for the initial detection of TB, with and without rifampicin resistance, among adults and adolescents who are unable to produce sputum. Tongue swabs are described as readily available and easy to collect, offering a practical option where conventional sputum collection is not feasible.
Sputum pooling: Pooling of sputum samples is now endorsed as a diagnostic strategy for initial detection of TB and rifampicin resistance using LC-aNAATs. The strategy has the potential to improve turnaround times and reduce costs in resource-constrained settings.
Closing the Diagnostic Gap
The recommendations come amid ongoing shortfalls in TB detection, even as countries have committed—under WHO’s End TB Strategy and the political declaration of the United Nations High Level Meeting on TB—to ensuring early diagnosis and universal access to WHO-recommended rapid molecular tests.
According to WHO, critical barriers continue to impede progress: sputum remains the dominant specimen type despite the fact that not all patients can produce it; laboratory-based tests are not always available at the sites where patients present for care; and the cost of testing equipment limits expansion of testing networks, particularly in low-resource settings.
What Comes Next
The full updated guidelines are expected to be published later in 2026. The release will be accompanied by the WHO Operational Handbook on Tuberculosis, Module 3: Diagnosis, 2nd edition, which will cover all currently recommended technologies, specimen types, and testing strategies, as well as step-by-step guidance for introducing new TB diagnostics into health programs and model algorithms for testing and clinical management.
WHO also plans to release a dedicated Toolkit for Near Point-of-Care and Swab-Based Tuberculosis Testing, which will include customizable planning, readiness assessment, training, and monitoring and evaluation materials to support program implementation. A series of regional webinars with WHO and partner organizations is also planned following the guideline release.
National TB programs and other stakeholders are encouraged to seek advice from WHO before introducing the new technologies recommended in the revised guidelines.
ID 417033865 © Wave Break Media Ltd | Dreamstime.com
Related Articles:
Co-Diagnostics Develops Sample Prep Instrument for Point-of-Care Tuberculosis Testing
CDC Selects Cepheid as National Collaborator for Rapid Diagnostic Development
The New Era of Respiratory Virus Testing