The partnership is designed to streamline targeted genomic testing for sponsors running biomarker-driven lung cancer trials.
CellCarta and Biofidelity have announced an expanded multi-year global strategic partnership that grants CellCarta exclusive rights to deploy Biofidelity’s Aspyre Lung assay in clinical trial settings worldwide.
Building on an initial collaboration announced in early 2025, the agreement is intended to give trial sponsors a single, coordinated partner for Aspyre Lung deployment, reducing the complexity of managing multiple organizations for the same platform.
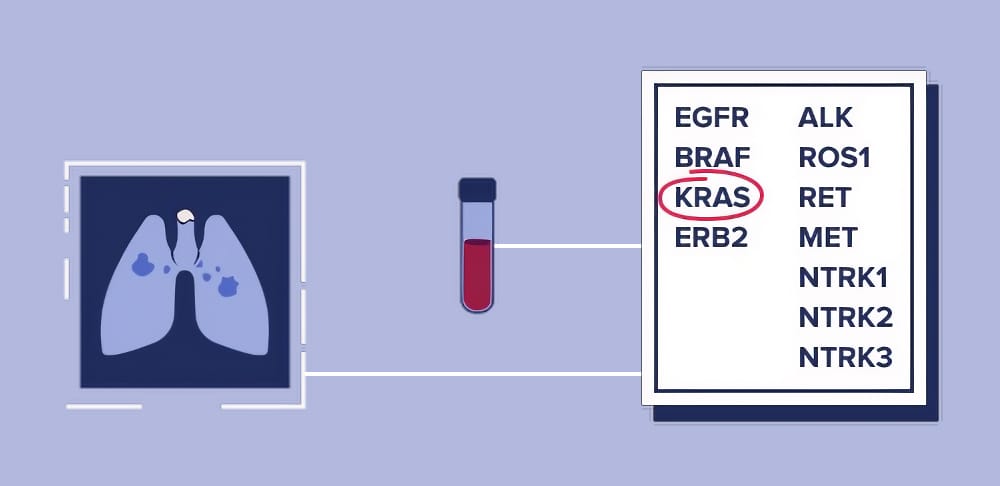
Aspyre Lung is a targeted assay purpose-built for lung cancer. It detects actionable genomic alterations in both tissue and blood samples while requiring minimal specimen input. According to the companies, the assay delivers results in four to five days, achieves a success rate as high as 99%, and is more cost-effective compared to commonly used broad next-generation sequencing (NGS).
The announcement comes as broad NGS panels—frequently used for biomarker-guided patient enrollment and treatment decisions in lung cancer trials—face scrutiny for their operational limitations. According to the companies, broad NGS panels can take three weeks or more to return results, and 25% of lung cancer samples fail NGS testing entirely.
“Becoming the exclusive clinical trial partner for Aspyre Lung is a significant evolution in our partnership with Biofidelity,” says Robin Grimwood, senior vice president of genomics at CellCarta, in a release. “With this agreement, we are establishing a more coordinated framework for deploying Aspyre Lung in global clinical trials and positioning ourselves well for future collaborations on emerging assays, such as Enspyre, Biofidelity’s minimal residual disease (MRD) technology.”
Reducing Operational Friction for Trial Sponsors
The expanded partnership is structured to streamline how targeted genomic technologies are integrated into global clinical studies. By centralizing assay expertise and clinical trial delivery under one partner, the agreement aims to reduce operational friction for sponsors incorporating targeted testing into their programs, according to the companies.
“This expanded partnership reflects our shared commitment to simplifying how targeted genomic technologies are integrated into clinical trials,” says Barnaby Balmforth, chief executive officer at Biofidelity, in a release. “CellCarta brings a proven track record of quality, reliability, and operational excellence in global clinical trial testing. Together, we aim to reduce barriers to adoption and enable sponsors to more easily incorporate targeted lung cancer testing into their trials.”
Photo caption: ASPYRE Lung identifies actionable genomic alterations across key lung cancer biomarkers — including EGFR, ALK, KRAS, BRAF, and more — from both tissue and blood samples, delivering results in 4–5 days. CellCarta now holds global exclusivity to deploy the assay in clinical trial settings.
Photo credit: CellCarta