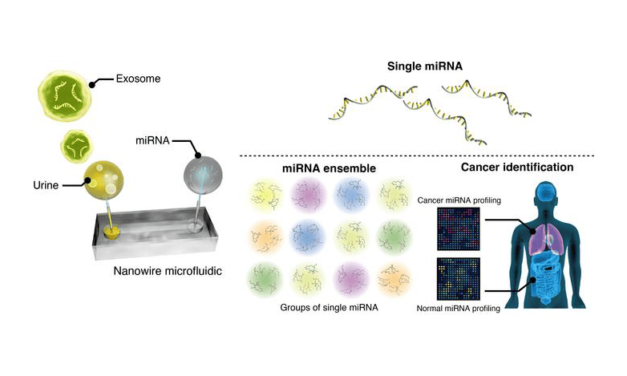
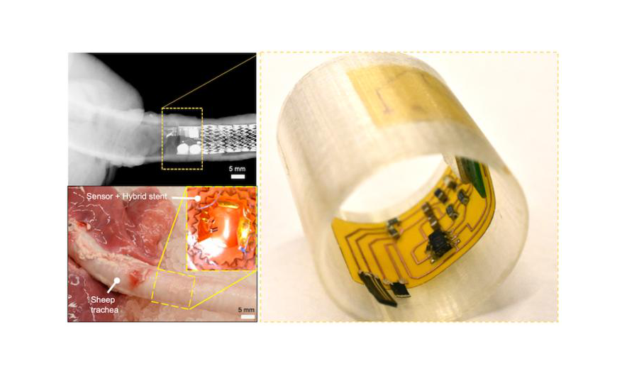
New Clinical Data Supports Blood-Based Monitoring for Cancer Treatment Response
Personalis presented data at AACR showing its NeXT Personal assay enabled ultrasensitive ctDNA monitoring for colorectal and lung cancer patients, demonstrating 100% sensitivity at baseline and 100% specificity for relapse post-surgery.