Scientists from Rice University have developed a noninvasive way to monitor gene expression dynamics in the brain, making it easier to investigate brain development, cognitive function, and neurological diseases, according to a study published in Nature Biotechnology.
Rice bioengineer Jerzy Szablowski and colleagues have engineered a unique class of molecules, known as released markers of activity (RMAs), that can be used to measure gene expression in the brain through a simple blood test.
“Typically, if you wanted to look at gene expression in the brain, you would have to wait to do a post-mortem analysis,” says Szablowski, an assistant professor of bioengineering at Rice’s George R. Brown School of Engineering. “There are some more modern neuroimaging techniques that can do this, but they lack sensitivity and specificity to track changes in specific cell types. With the RMA platform, we can introduce a synthetic gene expression reporter to the brain, which produces a protein that can pass through the blood-brain barrier. We can then measure changes in expression for a gene of interest with a simple blood test.”
Szablowski first considered the possibility of a synthetic gene expression reporter after noting the brain would quickly clear antibody therapy injections.
“Whenever these injections were done, the antibodies would just disappear⎯they wouldn’t hang around long enough in the brain for an effective therapy,” he says. “But we thought the failure of antibody therapies could be repurposed to our advantage. What if we took the part of the antibody responsible for this escape and attached it to a protein that could be easily detected? We could then see where, when and how much of a particular gene was being expressed in the brain.”
Other researchers had already determined that antibodies make their way through the blood-brain barrier using the neonatal fragment crystallizable receptor (FcRn), a gene known for helping maintain the level of antibodies present across the body. Using sophisticated bioengineering techniques, Szablowski and team attached the part of the antibody that helps it get through the blood-brain barrier to a common reporter protein to take advantage of this biological escape hatch. When the group then connected the RMAs to a specific gene and expressed that gene in the brain of a mouse, they were able to see that expression mirrored in the animal’s blood.
Further reading: Automated Liquid Biopsy Detects Brain Tumor Cells in Children
“This method is very sensitive and can track changes in specific cells,” Szablowski says. “Producing this protein in approximately 1% of the brain raised its blood levels up to 100,000-fold compared to baseline. We could specifically track the expression of this one protein with just a blood test.”
For now, Szablowski sees RMAs as a vital research tool to help scientists better monitor gene expression in the brain. For example, he says, the RMA platform could be used to look at how long novel gene therapies stay the brain over time.
“We could track these new therapies with just a blood test and continue to monitor them over time since the RMA platform is noninvasive,” he says. “But we can also use RMAs to study gene expression as it relates to disease. Being able to track different gene expression changes will allow us to understand what leads to disease and how the disease itself changes gene expression in the brain. This could provide new clues for drug development, or even for how to prevent neurological diseases in the first place.”
The David and Lucile Packard Foundation (2021-73005) and the National Institutes of Health (R21EB033059, DP2GM140923, R00DA043609, F31NS125927) supported the research.
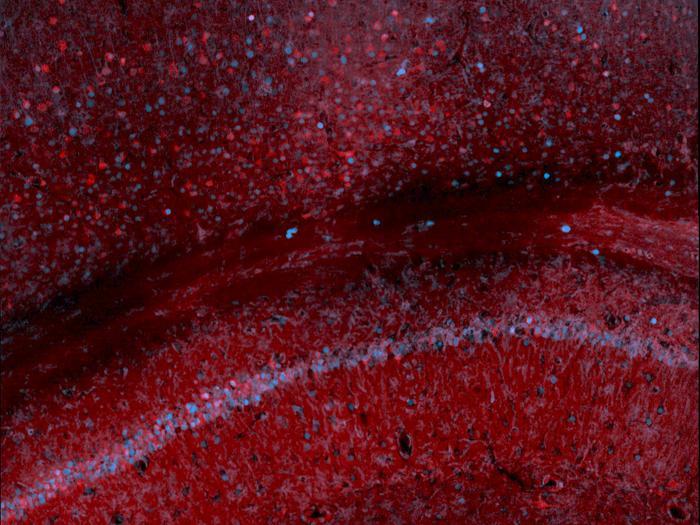
Featured image: Scan of mouse hippocampus and cortex, where the red signal derives from immunostaining for the synthetic marker developed by the Szablowski lab and the blue from the immunostaining of c-Fos, an endogenous protein whose production is heightened by increased neuronal activity. Photo: Szablowski lab/Rice University