By Nicholas Borgert
 More than 50 years ago, a research team in Scotland watched powerlessly as the conditions of two seemingly healthy infant siblings deteriorated. Within 9 weeks of their birth, the infants died.
More than 50 years ago, a research team in Scotland watched powerlessly as the conditions of two seemingly healthy infant siblings deteriorated. Within 9 weeks of their birth, the infants died.
Autopsies of the infant boy revealed massive cell inflammation in his liver and highly reactive marrow. The infant girl’s spleen showed extensive presence of histiocytes—many with erythrophagocytosis—as well as lymphocytes and polymorphs.
These conditions continue to signal a rare and often fatal disease known as hemophagocytic lymphohistiocytosis (HLH). In its primary form, HLH is genetically determined; the secondary form is acquired by the young patient and results from infections and malignancies. The disease threatens both genders. While HLH has been diagnosed in utero, in most cases its symptoms surface during the first 2 months after birth. Sometimes, HLH occurs in adolescents and young adults.
In both forms, patients typically suffer from a prolonged fever, jaundice, hepatosplenomegaly or enlarged spleen, and often a rash. This rare disease has spurred the formation of several important groups through the years. Physicians and researchers from around the world founded the Histiocytosis Society to pool knowledge of HLH and related immunodeficiencies. The Histiocytosis Association of America (HAA) has also been active.
Scientists do not yet know all of the complicated processes involved in the disease. Statistics about HLH are difficult to locate. A spokesman for the Centers for Disease Control and Prevention in Atlanta says the agency’s birth-defects center keeps no data on the incidence of HLH; information collected about HLH by the National Institutes of Health is limited.
A Rare Immunodeficiency
The most common figure cited for HLH incidence in the United States is one diagnosis for every 50,000 live births. That compares with other pediatric diseases such as galactosemia (1 in 62,000 births) and phenylketonuria (1 in 12,000 births), according to Kenneth McClain, MD, of the Baylor College of Medicine and Texas Children’s Hospital in Houston.
In Sweden, incidence of the disease is a mere 1.2 cases per 1 million children. Rates in Hong Kong and Taiwan are significantly higher but researchers haven’t determined why. HLH strikes patients most often during the summer months.
The human body produces many types of cells responsible for fighting off common and not-so-common infections. One type of infection-fighting cells—called histiocytes—roams throughout the body and anchors in various body tissue after being produced in human bone marrow.
Patients with active HLH bear an excessive amount of histiocyte cells and often lymphocytes, both of which are white blood cells. This excess can result in inflammation that produces swelling, redness, heat, pain, and loss of function.
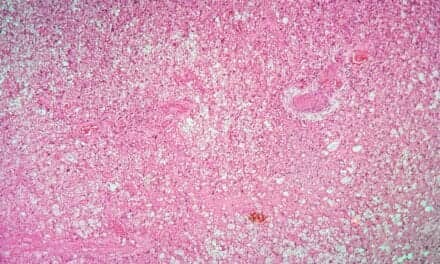
 Hemophagocytic histiocyte from biopsy of patient with HLH.
Hemophagocytic histiocyte from biopsy of patient with HLH.
These cells can quickly penetrate and settle in good tissue in many organs and body locations. These include bone marrow, lymph nodes, liver, spleen, skin, membranes around the brain, spinal cord, and sometimes even within the brain itself.
A Genetic Breakthrough
In 1999, researchers made the link between HLH and mutations in the perforin gene (PRF1), a gene responsible for helping generate natural killer cell (NK) functions. NK cells are critical to healthy immune systems. Involvement of a second gene is suspected but has not been documented. Secondary HLH tends to be diagnosed most often in older children, teenagers, and those with autoimmune diseases.
HAA President Jeff Toughill says survival rates for patients diagnosed with HLH have improved from zero a decade ago to nearly 60% now. The rarity and low public awareness about HLH has not stopped dedicated physicians and leading hospitals from achieving advancements in fighting the disease.
Diagnosing HLH
Kenneth L. McClain, MD, professor of pediatrics at Baylor College of Medicine, Texas Children’s Cancer Center/Hematology Service, has been involved in treating HLH since 1979 during his hematology/oncology fellowship at the University of Minnesota.
According to McClain, HLH can be triggered by many different kinds of infections or as an additional vulnerability when someone has juvenile rheumatoid arthritis, lupus erythematosis, or T-cell lymphoma. Also, it can occur in immune-suppressed patients who have an organ transplant or leukemia, he says.
Basic criteria for diagnosing HLH include fever, splenomegaly, a decrease in two blood cell counts (Hgb, white-cell count, or platelets), hypertriglyceridemia, hypofibrinogenemia or both, and hemophagocytosis in the bone marrow, spleen, or lymph nodes without evidence of malignancy. Sometimes, he says, a second marrow procedure is needed because up to 20% of patients cannot be diagnosed on clinical criteria alone.
More recent criteria include low or absent NK-cell activity or combination of serum ferritin of greater than 500 µg/L and soluble CD25 in excess of 2,400 U/ml. He says his facility uses the NK/perforin tests on all patients.
“Treatment with chemoimmunotherapy (HLH-94) is necessary to get the patient in remission,” McClain says. “For some this is sufficient; for others with familial disease or mutations in the perforin gene, a bone-marrow transplant is needed.”
A decade ago, a group of French physicians and researchers treated six HLH patients with antithymocyte globulin, steroids, and cyclosporine. The first reports indicated one patient died, two underwent stem-cell transplants, and three were kept on cyclosporine for 6 to 24 months and were in remission. It is unclear whether these patients were truly cured.
McClain says NK-cell function with perforin staining quantification by flow cytometry and perforin gene mutation analysis are among the newest tools available to clinicians for diagnosing HLH in patients. Also, magnetic resonance imaging (MRI) of the brain is used to look for enhancement of mass lesions. A lumbar puncture is needed for evaluating white-cell count, protein, and glucose levels.
“The HLH-94 treatment protocol now updated as the HLH-2004 protocol continues to be the most recognized and successful regimen for treatment of HLH patients,” says McClain. Despite a 60% “cure” rate, a lot of work remains to be done, he says.
“Much of this mortality relates to delay of treatment, but even with prompt institution of treatment, some patients die,” McClain says. “It is a very sneaky disease that is easily confused with others. I doubt we will stamp it out completely, but with continued research, we may find better targets for treatment.”

A Look Into the Future
Robert J. Arceci, MD, PhD, has been involved in treating children with HLH and other histiocytic disorders since 1981. The author of dozens of papers on the disease, a former faculty member at the Dana-Farber Cancer Institute and Boston Children’s Hospital, and the former director of Pediatric Hematology/Oncology at Cincinnati Children’s Hospital, Arceci today serves as director and King Fahd Professor of Pediatric Oncology at the Sidney Kimmel Comprehensive Cancer Center at the Johns Hopkins University School of Medicine.
According to Arceci, HLH is diagnosed in approximately 0.12 cases per 100,000 children each year. For comparison, the annual incidence of Langerhans cell histiocytosis, a related disease, has been calculated to be between 0.3 and 0.77 cases per 100,000.
“The currently accepted treatment of either primary or refractory/relapsing HLH is chemotherapy and immunosuppressive therapy followed by bone-marrow transplantation,” says Arceci. In the future, he says, therapies for HLH caused by known genetic defects may involve gene therapy directed at replacing the defective gene in hematopoietic stem cells as well as novel approaches to bone marrow transplantation.
“We are particularly involved in developing safer and more effective approaches of hematopoietic stem-cell transplantation for HLH and other inherited disorders,” Arceci says. “Some of these novel methods are designed to generate tolerance of the donor cells toward the host in order to avoid the very morbid and sometimes fatal effects of graft-versus-host disease.”
He anticipates that novel approaches to gene therapy and transplantation will improve the outcome for patients with HLH in the future.
“Targeted therapies directed more specifically at the out-of-control T-cells and macrophages should also be able to reduce the often severe side effects of current therapies,” Arceci says. “In addition, preclinical animal models of these disorders, in which new therapies can be quickly and effectively tested, should accelerate progress in our understanding and treatment of these diseases.”
Initial HLH-94 Protocol
An induction protocol known as HLH-94, pioneered by an international team of researchers, has become the most accepted way to treat newly diagnosed HLH. The HLH-94 protocol combines antihistiocyte and anti-T-cell therapies; the goal is initial clinical remission of the disease. This therapy includes doses of intensive etoposide and dexamethasone followed by a maintenance level of cyclosporin A.
A study published 2 years ago found most patients treated with HLH-94 showed significant symptomatic response within 2 weeks of the start of treatment. Overall results with HLH-94 cited in the study indicated a 55% survival rate at 3 years for patients.
Marrow Transplants
By far the most common treatment for HLH is the replacement of a patient’s immune system through bone-marrow transplants. The first published successful bone-marrow transplant for HLH occurred in 1986, using marrow donated by a sibling of the young patient.
A report 10 years later, using data from the International HLH Registry, indicated more than 60% of children who had received donated marrow from a sibling were alive 5 years later. (Only 10% of patients treated with chemotherapy were alive 5 years later.) The better news is that survival rates for patients using marrow from a nonrelated donor are nearly as high as those using sibling donors.
HLH Testing at Cincinnati Children’s Hospital Medical Center
Alexandra Filipovich, MD, is director of the Immunodeficiencies and Histiocytosis Program at Cincinnati Children’s Hospital Medical Center (CCHMC) and medical director for its Hematology/Oncology Diagnostics Laboratory. She is a board member of both the Histiocyte Society and Histiocytosis Association of America.
CCHMC offers initial diagnostic evaluations and ongoing management, home-based therapies, immunologic testing, and genetic diagnosis of many immunodeficiencies. Consultations for second opinions on immunodeficiency conditions are also available. The hospital participates in national trials involving the use of high-dose intravenous immune globulin (IVIG) as well as hematopoietic stem-cell transplants—from both related and unrelated donors of marrow, cord blood, and peripheral stem cells.
CCHMC also serves as national reference laboratory for immunologic testing of HLH. Filipovich and her colleagues have developed a rapid screening assay for a common genetic type of HLH due to perforin deficiency. The screening assay is available through referrals to pediatricians around the globe.
The hospital offers HLH testing with several goals in mind. Through tests, the hospital can exclude other possible conditions such as malignant diseases. Testing also can identify potential infectious cofactors that may need treatment and can determine the extent of the disease in patients. A valuable by-product of testing is the collection of material to help characterize possible genetic defects for future reference.
A typical diagnostic regimen involves a physical exam with complete patient history, complete blood count, coagulation profile (including fibrinogen levels, serum electrolyte and fasting triglyceride levels), diagnostic bone-marrow biopsy, and lumbar puncture. Collected samples include blood, marrow, cerebral spinal fluid, urine, and stool. Another common choice is quantitative PCR testing for Epstein-Barr virus and cytomegalovirus. Also, assisting in the diagnosis is the use of an MRI of the brain, which helps to establish a baseline for later comparisons.
To assist in diagnostics, CCHMC offers flow cytometry and cell sorting. The facility is equipped with two FACSCaliburs using cooled argon and red-diode lasers. The lab has two FACSVantage SE high-speed cell sorters with advanced optics. These high-speed multi-laser cell sorts perform at speeds up to 20,000 cells per second, with fewer than 5% aborts and more than 98.5% purity for most sorted fractions.
For cells not hearty enough to survive sorting, the hospital is equipped with a CloneCyt Plus machine. This enables fragile cells to be sorted onto glass slides or by using 96-well microtiter trays.
The use of flow cytometry to detect HLH caused by PRF1 mutations has proven highly reliable. The flow cytometry displays quantities of natural killer and other T-cells for comparison with age-related ranges. The hospital has found that the percentage of perforin-positive NK cells is usually greater than 85%—a percentage that is fairly stable regardless of age.
Writing in Immunology and Allergy Clinics of North America, Filipovich pointed out that while bone-marrow transplant can be expected to cure about two thirds of children with HLH, potential complications of that procedure can include severe inflammatory reactions, hemolytic anemia, and graft-versus-host disease.
“As with other rare conditions,” wrote Filipovich “the results of bone marrow transplantation are generally better when the procedure is performed at a major pediatric transplant center where the medical staff is familiar with the treatment of infants and patients with HLH.”
Nicholas Borgert is a contributing writer for Clinical Lab Products.