Infinium DNA Methylation BeadChip
High-density solution

The new Infinium DNA Methylation BeadChip from Illumina Inc, San Diego, is for use in epigenetic research. With this new high-density solution, researchers can assay approximately 27,000 CpG sites per sample, 12 samples at a time. The sites cover more than 14,000 well-annotated genes with single CpG resolution. Content for the panel was derived from genes described in the National Center for Biotechnology Information Consensus Coding Sequence Database, and features a core set of protein-coding regions that are consistently annotated and of high quality. The new array includes more than 1,000 cancer-related genes and 200 CpG sites located within CpG islands found in promoter regions of microRNA genes. The panel allows researchers to determine genome-wide methylation signatures.
Illumina Inc
(800) 809-4566
www.illumina.com
Technical Bulletin 5362
Shortening PCR run times
[register]
Bio-Rad Laboratories Inc, Hercules, Calif, is offering its new technical bulletin 5362, which describes how to shorten PCR run times by up to 50% using conventional PCR instruments, reagents, and reaction vessels. Many researchers in the life, pharmaceutical, and forensic sciences may believe that fast PCR is obtainable only through the purchase of specialized and faster-ramping thermal cyclers, but the new bulletin explains how time savings can be achieved by modifying thermal cycling conditions. The six-page bulletin provides information on shortening run times for standard PCR from approximately 90 to 35 minutes, amplifying long targets (1 to 20 kb) three to four times faster than standard protocols, and obtaining real-time quantitative PCR (qPCR) data with SYBR Green or TaqMan chemistries in less than an hour. Diagrams, tables, and graphics show how to improve run time for each of the steps involved in a conventional PCR run process, shorten the run time of real-time qPCR, and save time amplifying long targets. An addendum provides general considerations for fast-PCR, the rapid optimization strategy for fast-PCR, and troubleshooting tips. The bulletin is available from the company and may be downloaded from its Web site.
Bio-Rad Laboratories Inc
(800) 224-6723
www.bio-rad.com
Imubind ADAMTS13 ELISA
Assay
The Imubind ADAMTS13 ELISA from American Diagnostica Inc, Stamford, Conn, measures the level of ADAMTS13 protein in plasma. The assay is useful for the differential diagnosis of congenital TTP and as an aid in the diagnosis of acquired TTP. The ELISA kit includes anti-ADAMTS13 antibody precoated 96-well microtiter plate, standard, detection antibody, and positive control. The standard is calibrated against purified recombinant ADAMTS13, and quantitative results are expressed as ng/ml.
American Diagnostica Inc
(203) 602-7777
www.americandiagnostica.com
Seeplex STD/HPV Test
One-tube test

The new Seeplex STD/HPV test from Seegene, Rockville, Md, screens for Chlamydia trachomatis, Neisseria gonorrhoeae, and 14 human papillomaviruses (HPV) high-risk types and five HPV low-risk types in a single reaction tube. High-risk HPV types are detected in 99% of cervical cancers, and HPV types 16 and 18 cause more than 70% of cervical cancers. Individuals diagnosed positive for HPV are, on average, infected with up to five different types of HPVs. Seeplex offers multiplexing PCR technology that enables simultaneous multipathogen detection. It works in combination with automatic detection systems such as capillary electrophoresis, and delivers testing accuracy, efficiency, and cost-effectiveness.
Seegene
(866) 733-4949
www.seegene.com

Urine Monovette Tubes
Urine-collection products
Sarstedt, Newton NC, introduces two new Urine Monovette tubes to its line of urine collection products. The short 8.5-ml version is available for front-end automation systems, and the standard 10-ml size is offered in white to protect light-sensitive analytes. Both are printed with graduations, bar code label placement line, and writing block. The tubes aid in hygienic urine collection and testing without needles, adapters, or cups. They are manufactured from polypropylene and feature a leak-resistant screw cap for handling and transport.
Sarstedt
(800) 257-5101
www.sarstedt.com
Anti-NTB-A-PE and CD226 (DNAM-1)-PE
IOTest reagents

Beckman Coulter Inc, Fullerton, Calif, introduces two new IOTest reagents—Anti-NTB-A-PE and CD226 (DNAM-1)-PE. Both are monoclonal antibodies (mAbs) to cell surface receptors for natural killer (NK) cell studies, which provide important information in disease research. These new flow cytometry reagents are compatible with the company’s FC 500 series and Cell Lab Quanta SC systems, and other instrument platforms. NK cells are known to discriminate between normal and virus-infected or tumor cells, and they express different types of cell-surface functional receptors. Inhibitory NK receptors detect surface molecules specifically expressed on normal cells, and they transmit signals that spare these cells from attack. Activating NK receptors detect surface molecules that are upregulated in virus-infected and tumor cells, but powerless in normal cells. Anti-NTB-A-PE reacts with a member of a newly discovered family of NK receptors called signaling lymphocyte activation molecule, which are important functional receptors in lymphocytes and other leukocytes. In certain cases of NTB-A expression, downstream mechanisms are thought to be involved in induction of T cell costimulation and in regulation of B cell tolerance. IOTest CD226 (DNAM-1)-PE recognizes a putative cell-corruption surveillance receptor in NK and cytotoxic T lymphocyte cells. It may also play a role in transendothelial cell migration. The preformulated, standardized IOTest Anti-NTB-A-PE and CD226 (DNAM-1)-PE antibodies are provided in 50-test vials and are labeled for research use only.
Beckman Coulter Inc
(714) 993-5321
www.beckmancoulter.com
HerpeSelect Express HSV-2
Point-of-care serology test
Focus Diagnostics Inc, Cypress, Calif, introduces HerpeSelect Express HSV-2, a point-of-care serology test to aid in the diagnosis of herpes simplex type 2 in sexually active adults and pregnant women. The test is simple enough for use in clinics and other point-of-care settings where a rapid diagnosis can facilitate patient counseling, including treatment and lifestyle changes. It provides a result from a blood sample within 15 minutes. The test has received FDA 510k clearance, and is cleared for use in CLIA moderately complex laboratories. The company plans soon to apply for CLIA-waived status so the test can be used in less complex settings, such as physicians’ offices. The prevalence of herpes has increased dramatically in the last 3 decades because it is often transmitted without the knowledge of the infected individual or the uninfected partner. Transmission occurs 70% of the time in the absence of symptoms, and symptoms are often misdiagnosed. More than 50 million people nationwide are infected, and 90% are unaware they have the disease. Recent studies have shown HSV-2 infected individuals are at increased risk of acquiring HIV, the virus that causes AIDS.
Focus Diagnostics Inc
(800) 445-0185
www.focusdx.com
Liquichek Diabetes Control
Third-party control
Bio-Rad Laboratories Inc, Hercules, Calif, introduces Liquichek diabetes control, a liquid human whole blood-based third-party control for monitoring the precision of Hemoglobin A1C tests on commonly used methods and instruments. It provides an unbiased assessment, helps detect changes in assay performance, and improves the quality of test results. The product is available in three levels with a 1-mL fill size. The Unity Interlaboratory Program, with peer group comparison, is available for use with the product, as is Unity Real Time, a QC data management software system designed to improve the effectiveness of quality control processes.
Bio-Rad Laboratories Inc
(800) 224-6723
www.bio-rad.com/diagnostics
Fraction-Focus
Fractionates proteomes
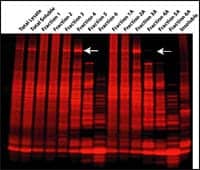
Fraction-Focus from G-Biosciences/Genotech, St Louis, relies on technology to fractionate and concentrate proteomes into multiple fractions, simplifying 2D maps and enhancing detection of low-abundant proteins. There is no detectable loss of material total protein during the procedure. At the end of the fractionation, cellular proteins are in one of many fractions. The entire fractionation is carried out in micro scale. The product is compatible with downstream protein identification techniques.
G-Biosciences/Genotech
(800) 628-7730
www.GBiosciences.com




