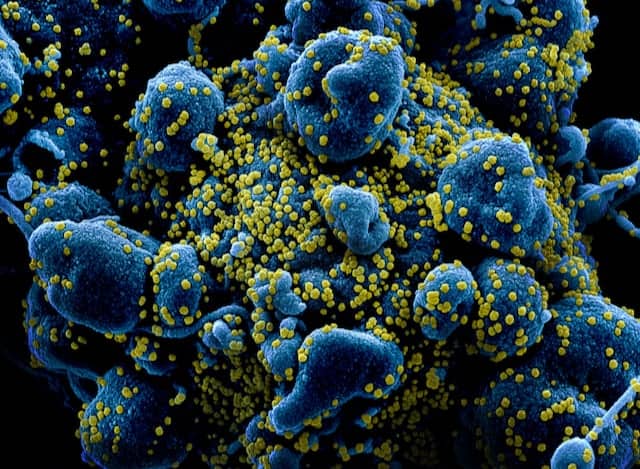
Veracyte, South San Francisco, Calif, and MaviDx, Miami, announced an agreement for MaviDx to develop an ultra-high-volume covid-19 test on the nCounter Analysis System, Veracyte’s diagnostics platform. The agreement is intended to enable genomic diagnostic testing and population screening for covid-19 at an unprecedented scale—up to 40,000 samples per day performed on the easy-to-use nCounter instrument—through technology that attaches molecular barcodes to individual RNA molecules of the virus. “The covid-19 pandemic has created a need for scalable diagnostic testing and population screening that we believe is not addressable with current technology, which is typically laborious and limited in capacity,” says Krassen Dimitrov, PhD, chief executive officer of MaviDx. “With our proprietary technology that allows patient results to be assessed from a highly multiplexed assay configuration, combined with the nCounter system’s proven accuracy and ability to perform high-throughput testing in an easy-to-use manner, we believe we can help address this growing global health need.” MaviDx is cofounded by Dimitrov, a founder of NanoString and inventor of the single-molecule barcode technology that powers the nCounter system. Veracyte acquired the exclusive global diagnostic rights to the nCounter system from NanoString in December 2019. Through the new agreement, MaviDx will develop, validate, secure regulatory approvals for, and commercialize its SARS-CoV-2 and other infectious disease tests, including for influenza, on the nCounter system. Veracyte has secured an equity stake in MaviDx and will supply the company’s infectious disease test kits and nCounter instruments to support laboratories and other entities in the United States and in global markets. Financial and other details were not disclosed. “We are pleased to partner with MaviDx and believe that their novel front-end technology, combined with the nCounter platform, has the potential to help make covid-19 testing widely available in clinical settings, as well as in the workplace, schools ,or other venues,” says Bonnie Anderson, Veracyte’s chairman and chief executive officer. “We look forward to supporting their efforts and to expanding our reach into this new area of significant unmet need.” MaviDx’s in-development test is intended to detect and quantify SARS-CoV-2 copy numbers from nasal or throat swabs, using the simple workflow of the nCounter system, with minimal manual handling. The technology being developed by MaviDx is designed to process up to approximately 9,000 patient samples at a time on a single cartridge, with the ability for a laboratory to potentially run up to five cartridges or more on the instrument during a 9-hour workday. For more information, visit Veracyte and MaviDx. Featured image: Colorized scanning electron micrograph of an apoptotic cell (blue) heavily infected with SARS-COV-2 virus particles (yellow), isolated from a patient sample. Image captured and color-enhanced at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID.
Veracyte, MaviDx Partner on Ultra-High-Volume Covid-19 Test