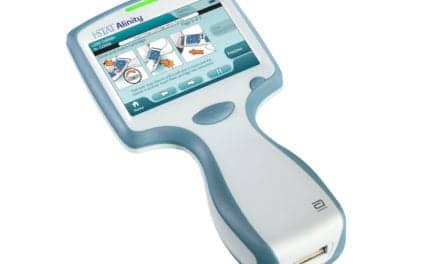
Diasorin received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the company’s new Liaison Plex platform as well as its first panel of tests, the Liaison Plex Respiratory Flex Assay.
The Liaison Plex is designed to provide full flexibility in the customization of syndromic panels and will allow clinical laboratory staff to overcome the constraint of the “one size fits all” approach for infectious disease syndromic diagnostics, the company says.
Custom panels allow for easier adjustment based on seasonal changes and clinical guidance updates as they occur, helping to avoid costly over-testing, which is a limitation of the fixed broad syndromic panels currently available to laboratories, Diasorin says. This fully automated, sample-to-answer system has a streamlined workflow with room-temperature stable consumables, allowing convenient storage and transportation. The operational hands-on time is only two minutes per sample, and results are produced in less than two hours.
“The LIAISON PLEX and its Respiratory Panel will allow clinical laboratories and physicians to tailor tests to the needs of their patient population,” says Angelo Rago, president of Luminex. “The LIAISON PLEX is truly a laboratory’s sample-to-customizable answer solution and we believe this will help address the growing demand for diagnostic stewardship, fiscal responsibility, and operational efficiency in clinical labs and healthcare systems throughout the U.S.”
The Liaison Plex is the successor of VERIGENE, which provides flexible solutions with its Respiratory Panel. The Plex system extends the Flex Testing and allows for full customization of its existing and upcoming panels, while incorporating full automation, Diasorin says. This minimizes sample preparation time and mitigates the risks of contamination for laboratory technicians. The enhanced flexibility and automation is designed to simultaneously add value for patients, clinicians, and payers.
The Liaison Plex will support Diasorin’s commercial strategy in the U.S. by targeting the hospital segment, leveraging IDNs, teaching institutions, and medical centers.
The Liaison Plex Respiratory Flex Assay tests for 19 pathogens commonly associated with respiratory infections, including 14 viral and 5 bacterial targets detected from nasopharyngeal swabs, further supporting flexibility and fiscal responsibility with a unique approach to panel testing. Flex testing also allows users to generate and pay for a subset of specific results based on a patient’s clinical picture.
Liaison Plex Respiratory Flex Assay is the first of the 5 panels announced during Diasorin 2023 Investor Day: Blood Culture panels (yeast, Gram-positive and Gram-negative) and Gastrointestinal panel are currently under development, and will be submitted to the U.S. FDA in 2024 and 2025, respectively.
Further reading: DiaSorin Simplexa COVID & Flu A/B Assay Gets FDA Clearance
Compared to standard panel testing, the Flex method can improve clinical utility and reimbursement rates, ensuring a more cost-effective approach for patients, the company says. Through the application of panel testing, patient care can be optimized by differential diagnosis and treatment in support of diagnostic stewardship efforts, thereby reducing unnecessary patient therapies and costly over testing.
“The clearance of the LIAISON PLEX along with its first panel marks a significant and strategic milestone for Diasorin after the Luminex acquisition, delivering to the market an innovative platform that is uniquely positioned in the growing multiplexing industry,” says Carlo Rosa, CEO of Diasorin. “The flex technology exemplifies our forward-looking approach to diagnostic development, tackling the current limitations of the multiplexing market with a fully automated and customizable panel, and providing customers with the needed flexibility to adopt this technology in a cost-effective manner.”