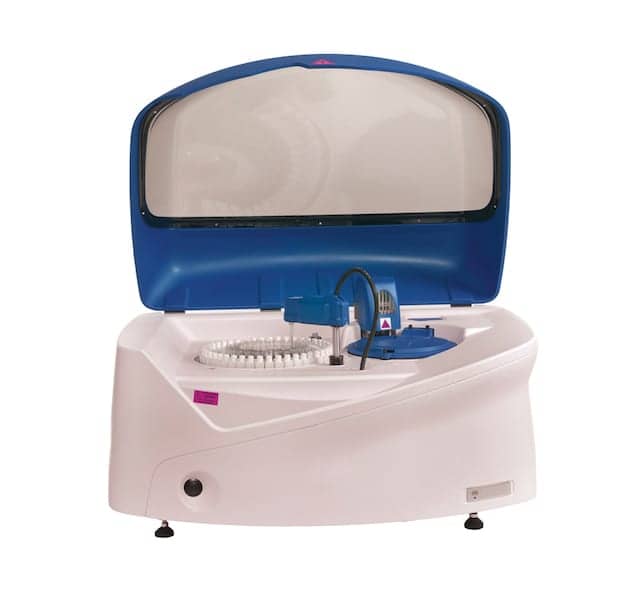
EKF Diagnostics, Cardiff, UK, recently launched the Altair 240 clinical chemistry analyzer in the European market. The system is already available commercially in the United States.
According to the company, the analyzer fulfills international demand for a fully automated, random-access, benchtop platform that is cost-effective as well as easy to learn, operate, and maintain.
Built to be compact and reliable, the Altair 240 is supported by a line of barcoded, liquid-stable, ready-to-use Stanbio chemistry reagents for routine and special chemistries. The analyzer includes positions for 43 reagents, and features the ability to configure open channels for esoteric testing, as well as stat interruption. As a result, the analyzer can be used as either a main or backup system, improving productivity in both hospital and physician’s office laboratory environments.
Incorporating laboratory information system bidirectional connectivity and running on intuitive Windows-based software, the analyzer automatically reads and manages barcoded reagent bottles and sample tubes. The system’s automated performance also enables auto-rerun and auto-dilution of samples.
Dual reagent probes translate to higher throughput and faster patient turnaround times. With an optional ion-selective electrode module, the system can perform up to 400 tests per hour. An integrated cuvette washing station enables the reuse of cuvettes. To minimize waste, the system’s continual 24-hour onboard cooling function ensures that most reagents have 30-day onboard stability.
For more information, visit EKF Diagnostics.