Inherited variants in the DPYD gene can increase the risk of life-threatening toxicity from fluoropyrimidine-based chemotherapies, yet screening remains inconsistent across care settings.
Colorectal cancer patients receiving fluoropyrimidine-based chemotherapies may face preventable, life-threatening toxicity when inherited variants in the DPYD gene go undetected before treatment begins—a risk that remains underscreened despite growing guideline support and updated safety labeling from the Food and Drug Administration (FDA).
Colorectal cancer is among the most commonly diagnosed cancers in the US and a leading cause of cancer-related death. Recent American Cancer Society data highlight a rising incidence in younger adults, reinforcing the urgency of earlier detection and more personalized treatment planning. For many patients, fluoropyrimidine-based therapies such as capecitabine and fluorouracil remain a cornerstone of treatment—but certain inherited variants in the DPYD gene can reduce dihydropyrimidine dehydrogenase (DPD) enzyme activity, significantly elevating the risk of severe or fatal toxicity.
The FDA has updated safety labeling for capecitabine and 5-FU to increase awareness of DPD deficiency risks and advises testing patients for genetic variants of DPYD prior to treatment initiation unless immediate therapy is necessary. Despite this guidance, screening remains inconsistent across care settings, leaving patients and providers without critical information that could affect treatment decisions.
“In precision oncology, treatment selection is not only about identifying what may work but also understanding factors that may affect how safely a patient can receive therapy,” says David Spetzler, president of Caris Life Sciences, in a release. “During Colorectal Cancer Awareness Month, it is important to recognize that DPYD testing can provide clinically meaningful information before treatment begins, helping clinicians make more informed decisions for patients who may be candidates for fluoropyrimidine-based therapy.”
Whole Exome and Transcriptome Sequencing as a Screening Tool
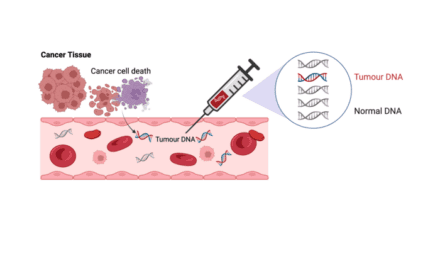
Caris Life Sciences addresses this gap by incorporating DPYD reporting into Caris Assure, its blood-based Whole Exome Sequencing and Whole Transcriptome Sequencing molecular profiling assay. The approach allows clinicians to assess both tumor biomarkers and clinically relevant inherited variants from a single blood draw, streamlining care and expanding the actionable information available before treatment begins.
By identifying inherited DPYD variants prior to therapy initiation, physicians may be able to reduce the risk of serious toxicity associated with fluoropyrimidine-based chemotherapies, according to the company.
A Broader Precision Medicine Conversation
The inconsistency in DPYD screening reflects a broader challenge in pharmacogenomic testing adoption across oncology. As the field advances toward more individualized care, awareness of pharmacogenomic factors—including DPYD status—is increasingly viewed as an important component of treatment planning in colorectal cancer.
Increasing education around biomarker-informed care may help support safer treatment planning and improve patient outcomes, according to Caris. The company notes that expanding access to integrated molecular profiling that captures both somatic and germline information from a single sample could help close the gap in pre-treatment risk assessment for patients with colorectal cancer.
ID 25092092 © Nikita Orlov Bernadskii
Related Reading:
Using Whole Transcriptome and Whole Exome Sequencing to Maximize Cancer Treatment
Quest Diagnostics Launches Pharmacogenomics Testing Service for Medication Guidance
FDA Approves Caris Life Sciences CDx for Targeted Cancer Therapy Precision
Guardant’s Shield CRC Blood Test Now Orderable Through Quest Diagnostics Network