Researchers have developed a new method of fluorescence microscopy for cell observation that circumvents fundamental problems of the practice: light-induced destruction and the unceasing movement of molecules.
This ultrarapid cryo-arrest of cells during live observation was developed in the Department of Systemic Cell Biology at the Max Planck Institute of Molecular Physiology in Dortmund, which now circumvents the issues.
Fluorescence light microscopy has the unique ability to observe cellular processes over a scale that bridges four orders of magnitude. Yet, its application to living cells is fundamentally limited by the very rapid and unceasing movement of molecules that define its living state.
Additionally, the interaction of light with fluorescent probes that enables the observation of molecular processes causes their very destruction.
The heart of the new approach is the cooling of living cells with enormous speeds up to 200,000 celsius per second to -196 celsius. This enables an unprecedented preservation of cellular biomolecules in their natural arrangement at the moment of arrest. This extremely fast, temperature cooling causes molecular movement to virtually stop.
Microscopic structures never stand still, but exhibit random as well as directed motion. Prolonging the exposure time thereby leads to blurring of the structures. Even more, the process of photon catalysis produces toxic radicals, which destroy molecular processes and eventually kill the cells, therefore limiting the amount of light that can be collected from the probes in the living cells.
Jan Huebinger, PhD, in the group of Philippe Bastiaens, PhD, developed the technology that arrests molecular activity patterns during observation of their dynamics in living cells at any timepoint of interest within milliseconds directly on the fluorescence microscope.
The researchers mastered this technical challenge by developing a ultrarapid cooling device that is integrated with a microscope where the cold of liquid nitrogen is accelerated under high pressure onto a diamond. The same diamond also holds the sample containing the cells on its opposing side.
The high pressure burst in combination with exceptional heat conductance of the diamond allowed to achieve the necessary high cooling rates to arrest cells at -196 celsius in their native configuration.
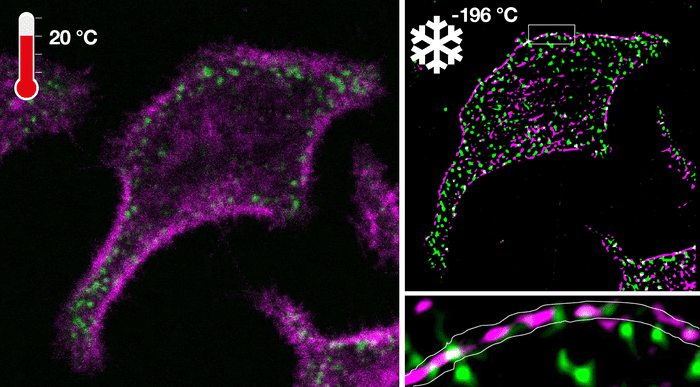
Ultrarapid cryo-arrest allowed the use of normally destructive high laser powers to analyze native molecular patterns at tens-of-nanometer resolutions that were otherwise invisible.
This new technology also led to the discovery of nanoscopic co-organization of an oncoprotein and a tumor suppressor protein that safeguards cells from exhibiting malignant behavior.
“This is an enabling step for fluorescence microscopy, especially the combination of super-resolution microscopy and microspectroscopy that allow the mapping of molecular reactions in cells at multiple scales. It will change the way we observe molecular organization and reaction patterns in cells and therefore provide more insight in the self-organizing capabilities of living matter,” says Bastiaens.
Featured Image: Fluorescence microscopy of an oncoprotein and corresponding tumor-suppressor in a living cell before cryo-arrest (left) and super-resolution image obtained under cryo-arrest (right). Photo: MPI of Molecular Physiology