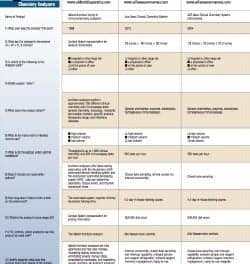
<%Response.charset="iso-8859-1"%>
Histos Series

Same-Day Diagnosis
Milestone Medical, Shelton, Conn, introduces Histos 3, Histos 5, and Histos Pro Rapid Microwave, designed to enable laboratories to achieve same- day routine histoprocessing with twice the productivity capabilities of conventional tissue processing. The Histo Series reduces processing times for transplant biopsies to 30 minutes, and other surgical biopsy tissue blocks of up to 5 mm thick to 4.5 hours, including fixation. These instruments can also perform decalcification, fixation, and special stains. The series enables laboratories to fulfill CLSI and CAP inspection guidelines.
Milestone Medical
(866) 995-5300
www.milestonemed.com
Shandon CheckMate

Verification system
Thermo Fisher Scientific, Pittsburgh, has introduced the Shandon CheckMate verification system, which is designed to eliminate sample mismatch errors in the anatomic pathology laboratory. This automated system can verify that sample containers, cassettes, and slides are matched. It eliminates sample mismatching errors by scanning and matching bar codes from slides, cassettes, and specimen containers at various workstations in the laboratory. Visual and audio indicators ensure that IDs are matched. This innovation improves result integrity in the lab by replacing error-prone, time-consuming manual checks, eliminating paperwork and identifying errors. The system, made up of bar code scanners, a mini PC, and proprietary CheckMate software, can be installed independent of the LIS, enabling slides and cassettes to be matched at workstations not connected to an LIS.
Thermo Fisher Scientific Inc
(800) 547-7429
www.thermo.com/pathology
Direct Enzymatic HbA1c

Eliminates Interference from Hemoglobin Variants
Diazyme Laboratories, San Diego, has announced the availability of its new liquid, stable, two-part Direct Enzymatic HbA1c assay, which can be used with a wide variety of clinical chemistry analyzers. Direct Enzymatic HbA1c requires only a single analyzer channel, compared to immunoassays that can require multiple channels. In addition, it eliminates the cuvette contamination that can occur with latex beads used in immunoassay methods. This method provides more reliable results than immunoassay or HPLC assays by eliminating interference from chemical and genetic hemoglobin variants. The product is certified traceable to the Diabetes Control and Complications Trial reference method.
Diazyme Laboratories
(888) 342-0963
www.diazyme.com
Maxwell 16 System

Integrated Personal Automation
Promega Corp, Madison, Wis, announced availability of the first fully integrated personal automation system in the United States that meets regulatory requirements for clinical diagnostic DNA purification. The Maxwell 16 System supports DNA-based tests performed by clinical labs, including sample preparation for human transplantation donor-matching tests done in HLA labs. With this product, up to 16 DNA samples can be purified simultaneously in about 30 minutes.
Promega Corp
(608) 274-4330
www.promega.com
Nova Max

No-Coding Blood Glucose Monitoring System
An advanced blood glucose monitoring system from Nova Biomedical, Waltham, Mass, provides a fast solution for diabetes self-testing. The newly released Nova Max is built around patented new technology test strips so precise that they do not require the calibration or coding required of currently available blood glucose monitors. Test results are reported in 5 seconds on a 0.3-µL sample. Results are accessed from the meter’s 400-test memory.
Nova Biomedical
(781) 647-3700
www.novabio.com