In critical care settings, tight glucose control is not just for diabetics
By Shara Rosen
Hypoglycemia and hyperglycemia may be complications of insulin therapy for patients in an intensive care unit (ICU). In the 1990s, several sentinel studies demonstrated the need for tight glycemic control for diabetic patients in critical care settings. The studies pointed to a correlation between variability in blood glucose levels and patient morbidity and mortality.
Studies headed by Anthony P. Furnary, MD, of St Vincent’s Hospital, Portland, Ore, led to the development of what is now referred to as the Portland Diabetes Protocols for tight glycemic control.1,2 Furnary’s publications were among the earliest to present the value of proper glycemic management for diabetic patients undergoing cardiothoracic surgical procedures such as coronary artery bypass graft surgery.
Following the research done by Furnary’s group, Greet Van den Berghe, MD, an intensive care specialist at the University Hospital of Katholieke Universiteit Leuven, Belgium, published findings on the application of intensive insulin therapy for critically ill patients in the New England Journal of Medicine.3 Those two researchers took the lead in the use of insulin titration for surgical intensive care patients.
Not long after the Furnary and Van den Berghe studies were published, commercial in vitro diagnostics companies began to demonstrate the ability of their point-of-care (POC) blood glucose meters to help in achieving tight glycemic control. However, most of those devices were a variation of products initially developed for glucose self-monitoring, with additional communication and information technology add-ons required for reporting and recording patient test results in a hospital setting. (For more information, see the companion article, “The Portland Diabetes Protocols.”)
Initially, none of those devices was specifically cleared by FDA for use in critical care environments, says Jeffrey A. Dubois, PhD, vice president for medical and scientific affairs at Nova Biomedical, Waltham, Mass. Nova Biomedical’s StatStrip glucose hospital meter system was the first blood glucose monitoring system to receive FDA clearance for use throughout all hospital and all professional healthcare settings, including intensive care settings.
Studies have also pointed to growing recognition of the need for continuous glucose testing for critical care patients—and especially in intensive care units—explains Richard Curtis, director of marketing at GlySure Ltd, Oxford, UK. GlySure anticipates CE marking in the near future for its GlySure continuous blood glucose monitoring (CBGM) system for use in continuous measurement of intravascular blood glucose.
“Over the past two decades there have been a number of studies done, and they all very clearly demonstrated that there is a definite improvement in morbidity and mortality in patients where tighter glycemic control was applied,” says Curtis. “And for tight glycemic control, the only way that you can apply this is by looking at the glucose levels on a continuous basis.” Generally speaking, he adds, hospitals do not have the infrastructure in place for nurses to take regular blood glucose measurements.
Furthermore, “for the last 15 to 20 years, there has been a fairly robust outpatient continuous blood glucose management market,” says Curtis. “That triggered our thinking that there is a need for continuous glucose monitoring in critical care, and there are proven continuous glucose monitors for glucose self-testing. When we added the scientific expertise of the Glysure development team, we put the three elements together.”
NEW REGULATIONS IN THE WIND
Most of the POC blood glucose meters used in hospitals were originally designed as self-monitoring devices, enabling people with diabetes to monitor the effectiveness of their glucose control programs through the use of fingerstick blood samples. Such devices have also become a tool for determining the glucose status of patients in hospitals, including those who are critically ill. The devices are CLIA waived, and present a tool that is easy to use at the patient’s bedside.
However, the ongoing trend toward greater use of ambulatory procedures means that the acuity of the patient mix in hospitals has increased. And a number of recent publications show that handheld bedside testing devices need improvement.4–7 According to Gerald J. Kost, MD, PhD, director of clinical chemistry and point-of-care testing in the department of pathology and laboratory medicine at the University of California, Davis, patients with complex treatment regimens are not always candidates for glucose measurement by means of a capillary fingerstick sample.
The use of POC glucose meters in the ICU is an area that has become particularly controversial because of one of its major uses—tight glucose control (TGC). The key concern is that current POC glucose meters may not be sufficiently accurate to meet the requirements of a TGC program. For this reason, FDA now requires that manufacturers include in their device labeling a notice that the product has not been evaluated on critically ill patients.
In turn, the use of POC glucose meters in other hospital settings has also come under scrutiny. In January 2012, the Clinical and Laboratory Standards Institute (CLSI) released new guidelines for the appropriate uses of POC blood glucose testing in the hospital, including performance standards for the glucose testing devices themselves.8
FDA has also become increasingly concerned about this practice, which it considers an off-label use, because critically ill patients may present with physiological variables that can interfere with the accuracy of blood glucose meters.
In January 2014, FDA released a draft guidance with suggestions for remedying this issue.9 In the past, the agency did not distinguish between consumer and professional uses of blood glucose monitors. With the release of two draft guidance documents—one for blood glucose POC devices used in clinical settings, and one for blood glucose POC devices manufactured for home use—FDA is now proposing that there be two sets of standards. Devices for professional and critical care use may be held to more rigorous standards given that the stakes are higher, as patients in such settings are fundamentally different from lay users. They may be acutely ill and medically fragile, so errors can lead to delays in insulin dosing and, in turn, to more episodes of hypoglycemia and conditions with unfavorable outcomes.
It has been suggested that FDA’s guidance for hospital use of POC glucose meters could have significant implications for device manufacturers and hospitals. If the draft guidance were to be fully implemented, POC blood glucose meters for professional use would be classified as moderate complexity devices under the terms of the Clinical Laboratory Improvement Amendments of 1988 (CLIA). Such a reclassification would change the requirements for device market clearance, and would also require healthcare providers using the professional meters to comply with stricter personnel and quality control standards.
In response to FDA’s draft guidance, the New York State Department of Health (NYSDH) informed the state’s hospital laboratory directors that POC glucose meters can no longer be used in critical care settings unless the device is used by an operator with high-complexity lab certification.10 The NYSDH policy also requires labs to perform studies showing that their blood glucose meters perform with an acceptable level of precision when used for off-label purposes. Until a lab meets these requirements, it must stop using glucose meters in critical care settings.
In an effort to calm the waters, in March 2015 the US Centers for Medicare & Medicaid Services (CMS) temporarily withdrew a November 2014 memorandum regarding off-label and modified use of CLIA-waived blood glucose monitoring systems, and announced a study period.11 “We seek to address some of these concerns, and clarify our long-standing requirements and policies where the feedback received thus far indicated that more education is warranted,” wrote the agency. “But we believe there is more work to be done. We plan to create additional forums for more discussion of these issues.” In the meantime, CMS invited stakeholders to submit comments via [email protected].
INDUSTRY DEVELOPMENTS
A 2009 study on glucose management headed by Simon Finfer, MD, of Royal North Shore Hospital, Sydney, Australia, also raised questions about the accuracy of POC glucose meters.12 “Because of the issues with POC devices, and uncertainty about the balance of risks and benefits of tight glucose control, Nova Biomedical designed the Normoglycemia in Intensive Care Evaluation–Survival Using Glucose Algorithm Regulation (NICE-SUGAR) trial to test the hypothesis that intensive glucose control reduces mortality at 90 days,” says Dubois.
Nova Biomedical developed a sophisticated biosensor strip designed especially for hospitalized patients. “We knew from the glycemic management protocol publications that there were confounding factors when certain blood glucose meters were used at the bedside,” says Dubois. “So we developed a sensor that measures hematocrit, and so can correct for hematocrit variance. And we also prepared the sensor in such a way that it can eliminate or significantly reduce any chemical interference from metabolites and drugs.”
In 2011, in cooperation with FDA, Nova Biomedical developed a protocol to study the impact of its device on bedside glucose measurement among critically ill patients. The study involved five university hospital-based centers—three in Europe and two in the United States—and a study population that involved 19 disease conditions based on the World Health Organization disease categories, and also represented 257 different medical conditions.
There were 1698 patients in the study, and 1815 measurements. The patients had been exposed to more than 8000 different drugs.
“The bottom line was that when we did a risk analysis of the device across the glycemic management control range, there were no clinically significant interferences,” says Dubois. “The results of the study were comparable to central lab test methods that were isotope dilution mass spectrometry traceable. This demonstrated that the device could be used across a broad spectrum of patient conditions.”
As a result, in September 2014 Nova Biomedical received FDA clearance to market the StatStrip glucose hospital meter system for the detection and management of dysglycemia throughout all professional healthcare settings, including critical care. The unit is also CLIA-waived in all settings. The company reports that the StatStrip device is the only glucose monitoring system that has this level of FDA market clearance. FDA originally cleared the device in April 2006 for use in hospitals as an aid in monitoring the effectiveness of a diabetes control program—but not for use with critically ill patients.
GlySure’s Curtis comments that several intravascular and subcutaneous blood glucose sensors have been launched in Europe, but they have not met with much success. One of the main reasons for this, he adds, is that they did not prove to be accurate when used across the whole of the patient portfolio. “Some products were more accurate than others, but even those that had good accuracy were limited when applied to certain patient groups.”
Curtis says that GlySure’s product development process has taken 8 to 9 years, and the company’s studies have involved some 358 patients to date. GlySure anticipates the company will be more successful than other companies. “This is because there are challenges that come with getting good accuracy across the whole portfolio, and that is something that GlySure has taken into consideration in developing the GlySure CBGM system.”
According to Curtis, GlySure’s studies have demonstrated that glucose concentrations measured by the GlySure system correlate with the concentrations measured in blood draws. “They concluded the system can dramatically reduce the need for frequent intermittent sampling, and that it offers the potential for early identification of hypoglycemic and hyperglycemic excursions across a broad spectrum of the patient population,” he adds.
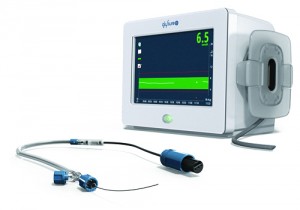
The GlySure continuous blood glucose monitoring (CBGM) system is expected to receive CE marking in 2015.
The GlySure CBGM system comprises two main parts: a monitor, and a sterile sensor set that includes an integrated fiber-optic sensor-introducer and a fully automated calibration module. According to Curtis, GlySure anticipates CE marking its GlySure CBGM system for intravascular continuous blood glucose.
GLYCEMIC STATUS FOR ALL PATIENTS
Hospitalized patients are very sick, and with illness comes a high degree of variability. So when manufacturers develop products, they should bear in mind that their devices should accommodate that variability. This factor was signaled by Alberto Gutierrez, PhD, director of the Office of In Vitro Diagnostics and Radiological Devices at FDA’s Center for Devices and Radiological Health, in a press release announcing the agency’s clearance of Nova Biomedical’s device for hospitalized patients. “It is important for manufacturers of glucose meters used in hospitals to design and test their devices for use in all hospitalized patients,” he said.
Nova Biomedical’s FDA clearance is for use in all areas of a hospital, and for indications that include using arterial or venous whole blood from patients with a wide variety of conditions, including cardiac, kidney, neurological, obstetric, gynecological, gastroenterological, endocrine, and lung issues; trauma; cancer; sepsis and infection; and people recovering from general or cardiothoracic surgery.
GlySure has taken this situation into consideration for its product development. “Over the past years we have noticed a change in the attitude of the medical community toward glucose and continuous glucose management,” says Curtis. “It is not just for critically ill patients, but also for general use in the hospital arena.”
Establishing glycemic control isn’t important only for diabetic patients, says Curtis, since any trauma may cause a dramatic rush in a patient’s glucose level. “We talk a lot about glycemic control in a diabetic patient,” he says. “But the idea of continuous glucose monitoring in the critical care setting is for the whole patient population—not just for diabetics.”
[reference id=”41545″]Sidebar: More Reading[/reference]
WHAT THE FUTURE MAY BRING
There is no doubt that there will be changes to glucose monitoring for hospitalized patients. Intermittent glucose measurement using POC glucose meters has been the prevailing tool for at least 40 years, but that method is coming under increased scrutiny.
“POC testing is not an excuse for inaccuracy,” explains Kost. “No matter which tests are performed in intensive care—whether for glucose or other analytes—their accuracy and precision should be as good as or better than traditional tests performed in a clinical laboratory.
“In fact, because of the elimination of delays in specimen processing, bedside testing will often be better,” Kost adds. “Certainly, in view of speed—that is, shortened therapeutic turnaround time—it will be more relevant to patient care.”
As for continuous glucose monitoring, “there are not a lot of published data to compare intermittent POC testing with continuous monitoring,” says Dubois. “There are some issues with continuous devices. Those that are subcutaneous work well with insulin pumps, but they are not necessarily recommended for glycemic control in hospitalized patients.”
Kost suggests that progress on continuous glucose monitoring has been substantial, but admits that numerous opportunities to advance biosensor and noninvasive detection stability and reliability still remain to be accomplished. “In the ICU, numerous challenges with performance have slowed progress,” says Kost.
Nevertheless, says Kost, continuous glucose monitoring still has great potential for use in critical care settings. “In intensive care, continuous monitoring with bedside alerts could help to eliminate episodic hypoglycemia, one of the main obstacles in caring for critically ill patients—and, of course, also for achieving tight glucose control.
“Continuous monitoring also has the potential to be used by patients as they transition from the hospital to home,” says Kost, “and it can be integrated with the in vivo administration of insulin through implants.”
When finally commercialized, the GlySure continuous glucose monitoring system will automatically record measurements and sound alerts at certain trigger points. By simply looking at a screen, says Curtis, the nurse will have a good indication of the patient’s glycemic state.
In the meantime, there are no arterial line devices for continuous glucose measurement cleared by FDA for use in the United States. Such devices would need to be calibrated, and might save nurses time, says Dubois. “But on the other hand, if you have a POC device that can produce a test result in 6 seconds, is it really necessary to have a continuous monitor that is intravascular?”
Shara Rosen is a contributing writer for CLP. For more information, contact CLP chief editor Steve Halasey via [email protected].
REFERENCES
- Furnary AP, Gao G, Grunkemeier GL, et al. Continuous insulin infusion reduces mortality in patients with diabetes undergoing coronary artery bypass grafting. J Thorac Cardiovasc Surg. 2003;125(5):1007–1021.
- Furnary AP, Wu Y. Clinical effects of hyperglycemia in the cardiac surgery population: the Portland diabetic project. Endocr Pract. 2006;12(Suppl 3):22–26.
- Van den Berghe G, Wouters P, Weekers F, et al. Intensive insulin therapy in the critically ill patients. N Engl J Med. 2001;345(19):1359 –1367.
- Punke MA, Decker C, Wodack K, Reuter DA, Kluge S. Continuous glucose monitoring on the ICU using a subcutaneous sensor. Med Klin Intensivmed Notfmed. Epub ahead of print, February 14, 2015; PMID: 25676120.
- Van Hooijdonk RTM, Leopold JH, Winters T, et al. Point accuracy and reliability of an interstitial continuous glucose monitoring device in critically ill patients: a prospective study. Crit Care. 2015;19(1):34; doi:10.1186/s13054-015-0757-4; Epub ahead of print February 5, 2015.
- Zhu W, Jiang L, Jiang S, Ma Y, Zhang M. Real-time continuous glucose monitoring versus conventional glucose monitoring in critically ill patients: a systematic review study protocol. BMJ Open. 2015;5(1):e006579; doi: 10.1136/bmjopen-2014-006579; Epub January 23, 2015.
- Sechterberger MK, van der Voort PH, Strasma PJ, DeVries JH. Accuracy of intra-arterial and subcutaneous continuous glucose monitoring in postoperative cardiac surgery patients in the ICU. J Diabetes Sci Technol. Epub ahead of print December 23, 2014; PMID: 25539652.
- Point-of-Care Blood Glucose Testing in Acute and Chronic Care Facilities; Approved Guideline. 3rd ed. [Guideline POCT12-A3.] Wayne, Pa: Clinical and Laboratory Standards Institute, 2012; available at: http://shop.clsi.org/point-of-care-documents/POCT12.html. Accessed March 19, 2015.
- Blood Glucose Monitoring Test Systems for Prescription Point-of-Care Use. [FDA draft guidance.] Rockville, Md: Center for Devices and Radiological Health, FDA, 2014. Available at: www.fda.gov/downloads/MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/UCM380325.pdf. Accessed March 19, 2015.
- New York State Department of Health. Directive Re: Off-Label Use of Glucose Meters. Albany, NY: New York State Department of Health, 2014. Available at: www.wadsworth.org/labcert/clep/files/Glucose_meters_off_label_use_1_13_14.pdf. Accessed March 19, 2015
- Reissuance of S&C 15-11 as Draft Only–for Comment Off-Label/Modified Use of Waived Blood Glucose Monitoring Systems (BGMS). Baltimore, Md: Centers for Medicare & Medicaid Services, 2015; available at: http://www.cms.gov/Medicare/Provider-Enrollment-and-Certification/SurveyCertificationGenInfo/Downloads/Survey-and-Cert-Letter-15-11.pdf. Accessed March 26, 2015
- Finfer S, Chittock DR, Su SY, et al. [The Normoglycemia in Intensive Care Evaluation–Survival Using Glucose Algorithm Regulation (NICE-SUGAR) study investigators.] Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009; 360(13):1283–1297; doi: 10.1056/NEJMoa0810625.