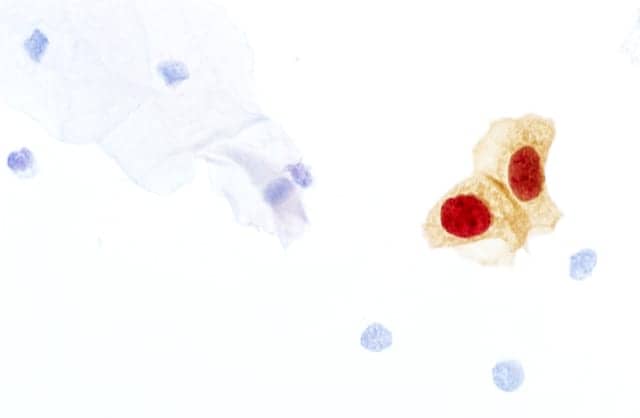
Roche, Basel, Switzerland, announced FDA approval for the expanded use of CINtec Plus Cytology, the first triage test based on biomarker technology for women whose cervical cancer screening results are positive for high-risk types of human papillomavirus (HPV). Additional information from this test supports clinical decisions about which women will benefit most from immediate follow-up. Laboratories can now use CINtec Plus Cytology to triage positive results from the cobas HPV Test run on the fully integrated, automated, and high-throughput cobas 6800/8800 Systems. High-risk HPV is the principal cause of cervical cancer, which is one of the most common cancers in women globally. The World Health Organization has set goals for countries to take action to eliminate cervical cancer by 2030. “With our portfolio of cervical cancer tests and automated testing platforms, we are committed to providing clinicians and laboratories with the best tools possible to protect women’s health,” says Thomas Schinecker, CEO of Roche Diagnostics. “This expanded indication for CINtec Plus Cytology gives laboratories the flexibility to triage cobas HPV test results on their choice of cobas Systems and deliver accuracy needed to reliably detect HPV infections that are starting to cause cellular changes that could lead to cancer. The biomarker information helps to clarify a woman’s risk of disease, reduce the potential for over- or under-treatment, and is a major step forward in individualizing a woman’s care.” HPV DNA screening identifies women at risk for cervical cancer by detecting the presence of high-risk HPV DNA in cervical samples. While most HPV infections resolve on their own, some women who test positive for the virus, or whose co-testing results are discrepant (HPV positive/Pap cytology negative), may have or may develop pre-cancerous cervical lesions. These lesions could progress to cervical cancer if left untreated. For more information, visit Roche.
FDA Approves Expanded Use of CINtec Plus Cytology