Today, the Centers for Medicare and Medicaid Services (CMS) released new tools to reduce burdensome paperwork and authorization delays for laboratories seeking Clinical Laboratory Improvement Amendments (CLIA) certification to test for coronavirus disease 2019 (covid-19). CMS’s quick-start guide helps laboratories with the application process for CLIA certification and includes information on the expedited review process implemented at the beginning of the public health emergency that allows labs to start covid-19 testing before the official paper certificate arrives by postal mail. Laboratories also have a new option to pay CLIA certification fees on the CMS CLIA Program website. Online payments are processed overnight, which is substantially faster than hard-copy checks. Today’s actions are part of the Trump Administration’s efforts to expand testing in the nation
“At President Trump’s direction, CMS has left no stone unturned in helping fight this highly contagious, dangerous disease,” says CMS Administrator Seema Verma. “An obscure process and outdated modes of payment have too often caused needless delays in certifying lab testing facilities. Today’s announcement will allow testing laboratories to promptly and painlessly register with CMS so they can get to work, focusing on providing reliable information to combat the spread of this disease.”
CMS regulates all laboratory testing performed on humans for the purposes of diagnosis, prevention, or treatment in the United States through the CLIA program. To become CLIA-certified, laboratories must meet performance and quality assurance requirements aimed at ensuring they are able to deliver reliable and accurate test results for the purpose of proper diagnosis, prevention and treatment of diseases like covid-19. This new guide provides laboratories with the resources they need to reduce paperwork and streamline the CLIA application and certification process. This quick-start guide outlines the steps laboratories must follow to apply for and receive CLIA certification, including ensuring the form is submitted to the correct state agency.
Prior to receiving certification, laboratories must also pay a user fee to cover the costs of administering the CLIA program, which also includes inspection costs. Laboratories can now pay CLIA certification fees through a secure platform hosted by the Treasury Department on the CMS CLIA Program website. CLIA fees are based on the certificate requested by the laboratory (i.e., Certificate of Waiver, Provider-performed Microscopy, Accreditation, or Compliance) and, in some instances, the annual volume and types of testing performed. The CLIA Certificate Fee Schedulecontains detailed information on costs. The actions announced today support continued efforts by CMS to promote access to safe and reliable testing during the COVID-19 public health emergency.
Read more from CMS.
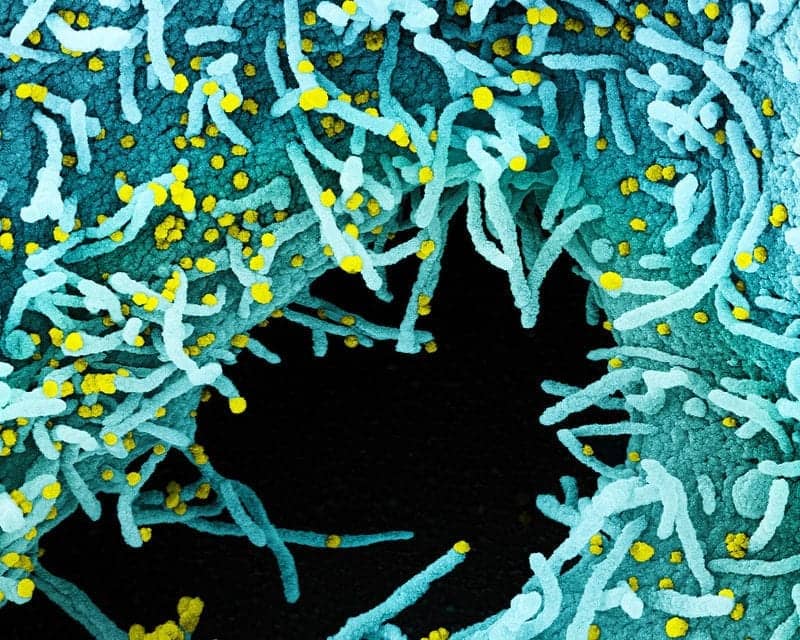
Featured image: Colorized scanning electron micrograph of a cell heavily infected with SARS-CoV-2 virus particles (yellow), isolated from a patient sample. The black area in the image is extracellular space between the cells. Image captured at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID