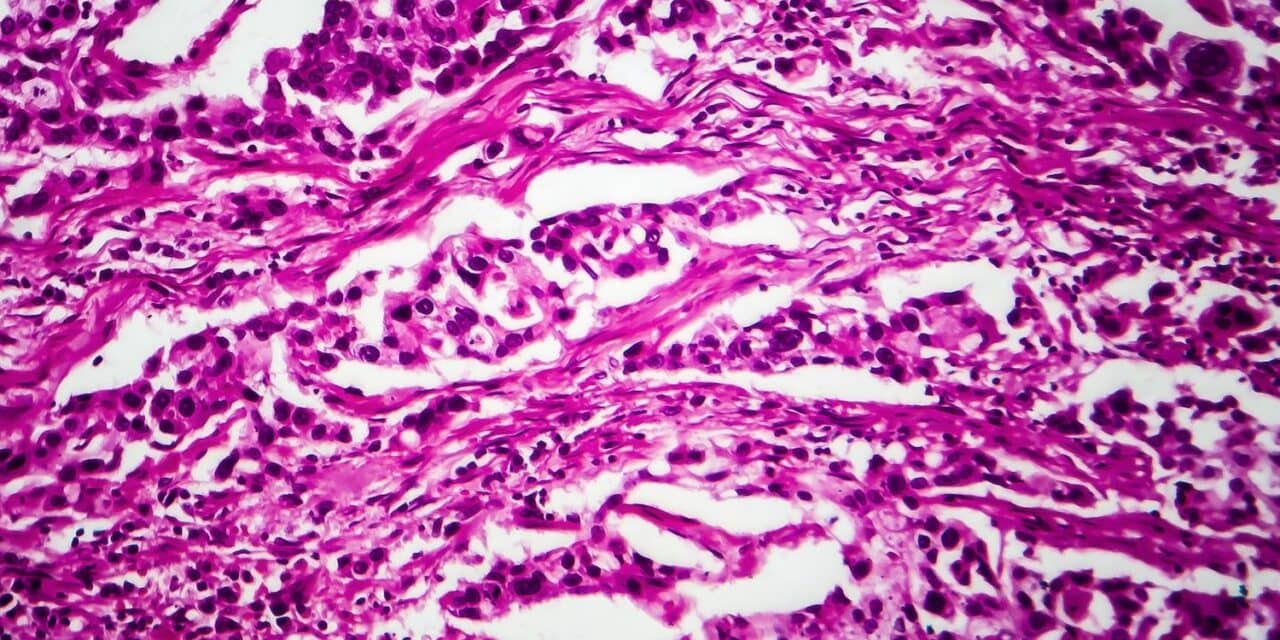
Los Angeles-based Nonagen Bioscience Corp, developers of liquid biopsy tests for the detection of human cancers, has announced Oncuria, their non-invasive bladder cancer test capable of predicting response to therapy, has been granted U.S. Food and Drug Administration (FDA) Breakthrough Device Designation.
This designation is granted to technologies that have the potential to provide more effective treatment, diagnosis or prognosis of life-threatening diseases, such as cancer. The designation enables close collaboration with, and expedited review by the FDA, and provides formal acknowledgement of Oncuria’s potential clinical benefit as a bladder cancer test.
Up to 50% of early-stage bladder tumors treated with surgery will recur. Guidelines for the treatment of early-stage bladder cancer recommend the instillation of bacillus Calmette-Guerin (BCG) into the bladder. BCG is an attenuated tuberculosis vaccine that acts as a non-specific immune system stimulant and as such is one of the first immunotherapies to treat human cancer. Nonagen says BCG has proven to be a successful treatment against residual bladder cancer; reducing recurrence rates and decreasing the progression to more advanced cancers. However, more than half of patients who receive BCG fail to respond. In 20% of patients, the disease grows and extends during or after BCG, which portends a worse survival. A diagnostic bladder cancer test performed prior to BCG treatment—capable of predicting treatment response—could be leveraged to avoid a futile treatment regimen. This could also avoid potential delays in the initiation of more appropriate clinical management which the company believes will benefit both patients and the healthcare system.
Oncuria, a multiplex immunoassay bladder cancer test, measures 10 protein biomarkers in a single voided urine sample. Biomarker levels are combined in a weighted algorithm to aid in the prediction of responding to BCG in patients with intermediate to high-risk, early-stage bladder cancer.
“Our algorithm is designed to provide prediction of response to BCG therapy, allowing for timely interventions that could result in more favorable outcomes for our patients,” says Charles J. Rosser, CEO of Nonagen. “We are proud that the FDA has decided to grant Breakthrough Device Designation to our lead diagnostic, Oncuria, acknowledging growing recognition of the benefit our test can offer to clinicians and patients.”