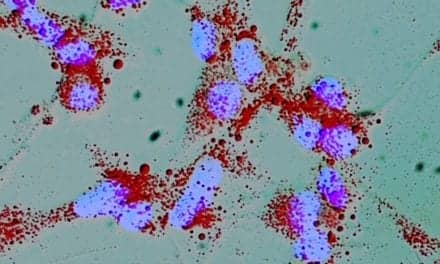
PSA test for prostate cancer should be repeated before biopsy
The prostate specific antigen (PSA) blood test is used in routine prostate cancer screening for men over 50, and elevated PSA levels typically result in referral for a prostate biopsy. In a study published in the May 28 Journal of the American Medical Association, researchers from Memorial Sloan-Kettering Cancer Center and colleagues suggest that biopsy should not be performed until the test is repeated, because PSA levels experience common fluctuations.
The study followed nearly one thousand men over four years, with results from five consecutive PSA tests. Up to one third of these men had elevated PSA levels that would usually result in a referral for a prostate biopsy, but subsequent testing a year or more later indicated that the PSA levels for half of the men had returned to normal.
“A single, elevated PSA level does not automatically warrant a prostate biopsy,” said Dr. James Eastham in a statement released by Memorial Sloan-Kettering. Dr. Eastham is a surgeon in the Department of Urology and the study’s lead author. “We recommend having the findings confirmed by repeating the PSA test after waiting at least six weeks. Even if the repeat test shows an elevated level, prostate cancer will only be discovered in about one quarter of men who undergo biopsy,” he said. Waiting to confirm the diagnosis will not have an adverse affect on those men who actually have prostate cancer, according to Peter Scardino, MD, chairman of the Department of Urology at Memorial Sloan-Kettering. “A delay in diagnosis of a few weeks or months is unlikely to alter treatment efficacy,” said Dr. Scardino. s
The retrospective study used annual blood samples that were stored and later analyzed for natural variations in PSA levels. While 21 percent of men had an elevated PSA (>4.0 ng/ml) at some time during the four year period, the level spontaneously returned to normal in nearly one half of the men and remained normal for most of them on further tests. A prostate biopsy of these men would not have been necessary.
“These natural variations in PSA detract from its use as a screening tool,” said Colin Begg, Ph.D., chairman of the Department of Epidemiology and Biostatistics at Memorial Sloan-Kettering and senior author of the study. “A policy of confirming newly elevated PSA levels several weeks later may reduce the number of unnecessary procedures as well as the number of men diagnosed with a small, incidental tumor that poses no threat to life or health.”
Other cancer specialists have suggested that physicians may need to pay closer attention to patients whose PSA levels fluctuate, as this variation may itself be cause for concern.

Working drafts of the honeybee and chimpanzee genomes are nearing completion, and scientists are set to begin the sequencing of other important model organisms, including man’s best friend. The boxer dog was chosen after analyses of 60 breeds because it has among the least amount of variation in its genome, increasing the likelihood of producing a reliable reference genome sequence.
According to the director of the National Human Genome Research Institute (NHGRI), Francis S. Collins, M.D., Ph.D., “Comparing the human genome sequence with those of other organisms helps us to identify regions of similarity and difference, providing critical clues about the structure and function of human genes. This information should point us toward better strategies for treating and preventing human disease. With each genome that we sequence, this approach becomes more powerful.”
The project to sequence the genome of the domestic dog (Canis familiaris) is part of NHGRI’s effort to support the rapidly growing field of comparative genomics. All dog breeds are more than 99 percent identical at the DNA level. The dog genome project, which is expected to cost about $50 million, will get underway in early June at the Whitehead/MIT Center for Genome Research in Cambridge, Mass.
A long history of selective breeding has left many types of dogs prone to genetic diseases like cancer, heart disease, deafness, blindness and autoimmune disorders that are very difficult to study in humans. The dog is also an important model for the genetics of behavior.
“Once we are able to compare the dog genome with the human genome, the dog will likely prove to be man’s best friend in more ways than we ever imagined,” said Dr. Collins, who notes that the genome sequencing effort will also benefit dogs by helping veterinary researchers develop new and better ways of combating canine diseases.
In its press release on the subject, the NIH notes that the dog genome is similar in size to the genomes of humans and other mammals, containing an estimated 2.8 billion
DNA base pairs. It is expected that the Whitehead/MIT team will complete a high-quality draft of the dog genome sequence within the next 12 months. After the genome of the boxer is sequenced, researchers plan to sample and analyze DNA from 10 to 20 other dog breeds to study genetic variation within the canine species.
In April 2003, the International Human Genome Sequencing Consortium announced the successful completion of the Human Genome Project. Researchers also sequenced the genomes of a number of organisms commonly used in biomedical research, including a Escherichia coli, baker’s yeast, two types of roundworm, Drosophila melanogaster, two types of sea squirt, two types of puffer fish, the mouse and the rat. By June of this year, sequencing work is expected to be completed for an initial working draft of the chimpanzee genome, using the human genome sequence as a guide.
“The chimp genome sequence will be important for many reasons, including the fact that it is the organism that is genetically closest to humans,” said Richard K. Wilson, Ph.D., director of the Genome Sequencing Center at Washington University School of Medicine.
Identifying the genes that seem unusually divergent between humans and chimps may prove extremely helpful in understanding disease susceptibility. For example, even though the chimpanzee DNA sequence is 98.8 percent identical to that of the human, chimps do not suffer from some diseases that strike people, such as AIDS and malaria. A more detailed comparison of the sequences of the two genomes may reveal that there are genetic reasons for such differences, suggesting new approaches for preventing and treating human disease.
| Updated practice guidelines may widen use of FISH testing for HER-2 The National Comprehensive Cancer Network (NCCN) has issued updated Practice Guidelines in Oncology that include new recommendations for testing HER-2 status in women with invasive breast cancer. The 2003 guidelines call for IHC and/or FISH testing as part of a breast cancer recurrence work-up, or initial work-up for stage IV disease. The guidelines go on to state that an IHC result of 2+ should be confirmed by FISH. HER-2 test results aid in the determination of whether a patient might benefit from treatment with the monoclonal antibody Herceptin (trastuzumab). Herceptin specifically targets tumor cells that overexpress the HER-2 protein. In 25 to 30 percent of breast cancer patients, the presence of multiple copies of the HER-2 gene on a single chromosome, called “gene amplification,” leads to overexpression of HER-2 protein, which plays a pivotal role in the rapid growth of tumor cells. Fluorescence in situ hybridization (FISH) testing measures the number of copies of the HER-2 gene at the DNA level. Using fluorescent coloring and a microscope, a diagnostician can count the actual number of HER-2 genes present in the cell nucleus. The most commonly used test method, immunohistochemistry (IHC), measures protein overexpression, which does not always correlate with increase in copy numbers of the HER-2 gene as determined by FISH. In IHC testing, which is more widely used, lower cost and less labor-intensive than FISH, the amount of HER-2 protein in a tumor is measured using a scale from 0 (negative) to 3+ (strongly positive). According to the National Cancer Institute, patients whose tumors are strongly positive for HER-2 protein overexpression (a score of 3+ on the laboratory test) are more likely to benefit from Herceptin treatment. There is no evidence of benefit in patients whose tumors do not overexpress HER-2 (a score of 0 or 1+). Tumors with IHC 2+ scores are considered borderline and, therefore, subject to interpretation. Several factors may also influence IHC results: among those noted have been reagent variability, time, processing techniques, etc. According to the new guidelines, FISH or IHC may be used in initial screening, then a FISH+ result should be used to identify likely responders to treatment once a patient has been identified as IHC 2+ or 3+. The Vysis division of Abbott Laboratories manufactures PathVysion, a FISH-based test the U.S. Food and Drug Administration (FDA) has approved for inclusion in the Herceptin package insert for the selection of patients who may respond to treatment. In addition to Herceptin therapy selection, PathVysion is also used for assessing patient prognosis and response to Adriamycin-based therapies. “Personalized treatment begins with an accurate diagnosis,” said Mark Pegram, M.D., associate professor of medicine, Division of Hematology/Oncology, and director of the Women’s Cancer Program, UCLA/Jonsson Comprehensive Cancer Center in a statement released by Abbott. “We rely on PathVysion to determine a woman’s HER-2 status because it reduces false negatives and false positives, clearly differentiating those patients who will most likely respond to Herceptin from those who may not. By using PathVysion, physicians can make better treatment decisions that may extend a patient’s survival.” |

A research letter to The Lancet published May 23, 2003 estimates that the prevalence of chlamydia infection in young men could be higher than previously thought, with up to 10 percent of young men affected.
The sexually transmitted chlamydia infection rarely leads to serious complications in men, but in women untreated infection can cause pelvic inflammatory disease and subsequent infertility. Chlamydia is often silent — research has indicated that half of the people affected are unaware of their infection. Screening among young women is now encouraged.
Nearly 90 percent of the 9.8 percent of young men found in the U.K. study to have chlamydia were asymptomatic. According to Dr. Gordon R. Scott, the lead investigator of the study, this much higher rate of asymptomatic infection than usually cited (90 compared to 50 percent), shows the importance of involving men as well as women in opportunistic testing for chlamydia.
Roche will transfer back Sysmex hematology business
Roche Diagnostics of Indianapolis, Ind., and Sysmex of Long Grove, Ill. have signed an asset purchase agreement to begin the transfer of Roche’s U.S. hematology business unit back to Sysmex. This agreement accelerates the expiration of an original Distributorship, Sales and Service (DSS) agreement that began in February, 1999, and was due to expire in 2004. Agreements for Roche’s distribution of Sysmex hematology outside the United States remain unchanged.
“Roche and Sysmex have enjoyed a good business relationship and progress has been made to gain market share in the very competitive hematology business. Although hematology is an element in the diagnostics market, it was decided that both Roche and Sysmex would achieve greater success and profitability by focusing on their respective strengths. Therefore, both parties agree that for business reasons, it is best that the business be transitioned back to Sysmex,” said Martin Madaus, Roche Diagnostics President and CEO.
“This pending move is in line with Roche’s strategy to increase our focus on the tremendous growth opportunities we have in diabetes, molecular diagnostics, immunochemistry and chemistry. The high growth rate we are experiencing in these core businesses will drive even more sales growth and improve profitability,” Madaus added.
“Sysmex is increasing our investments in the United States which will result in profitable market share growth,” said Kazuya Obe, Sysmex Chairman and CEO . “Sysmex’s core competencies and investments in diagnostics, including hematology, urinalysis and information solutions have resulted in a product portfolio that uniquely addresses not only clinical performance and efficiency, but integration of systems as well.”
Sysmex is currently expanding its operation by increasing hiring and continuing its planned move to larger headquarters.
| Roche to begin clinical trials for WNV blood center screening test Roche Diagnostics’ molecular division in Pleasanton, Calif., has received FDA authorization to begin clinical trials of the TaqScreen West Nile Virus Test, a PCR-based nucleic acid system that fully automates the specimen processing, amplification, and detection steps in blood screening. According to Roche, the company accelerated development of the test to address concerns expressed by the FDA and Centers for Disease Control and Prevention (CDC) last September about the potential for an increase in transfusion-related West Nile Virus infections during the 2003 mosquito season. Roche has now received FDA authorization to begin screening newly donated blood (versus archived samples) at U.S. blood centers. The company’s clinical trials are scheduled to begin in the United States and Canada (where Roche has filed a separate clinical trial application) on or before July 1. Eleven blood testing laboratories will screen samples from a network of nearly 100 local donation centers across the United States, including Alaska and Hawaii. Roche’s non-clinical (i.e., archived) studies have shown that, in addition to West Nile virus (Kunjin and New York strains), its test can detect other potentially deadly members of the Japanese Encephalitis virus group, including St. Louis Encephalitis virus, Japanese Encephalitis virus, and Murray Valley Encephalitis virus. A subset of the blood donations that test positive in the company’s trials for West Nile virus will be sent for further laboratory testing to determine the specific member of the Japanese Encephalitis group virus infecting the blood. |

The Howard University College of Medicine (HUMC) in Washington, D.C., in partnership with First Genetic Trust, Inc. (FGT) will develop the GRAD (Genomic Research in the African Diaspora) Biobank, a resource for genomics and clinical research in individuals of African descent. According to Howard, the Grad Biobank will enable the clinical genomic research community to advance understanding of the genetic and biological bases for differential disease risk, disease progression and drug response.
“Howard University is especially pleased to be partnering with First Genetic Trust on this groundbreaking initiative,” said Floyd J. Malveaux, M.D., Ph.D., dean of HUCM. “The GRAD Biobank will provide an infrastructure that will enable Howard University and the research community to conduct urgently needed research on health issues that greatly affect the well-being of people of African descent. Our collaboration with First Genetic Trust will allow us to accelerate the development of the GRAD Biobank, using the most advanced technologies available.”
Georgia M. Dunston, Ph.D., chairman of microbiology and director of the National Human Genome Center (NHGC) at Howard said, “Howard University has historically been a vanguard of the African and African-American community trust and is therefore the most appropriate institution to develop this initiative. In addition to producing and training leading physicians and health professionals, developing it will allow our students and faculty to participate in and develop expertise in medical technologies of the future while safeguarding the privacy and ensuring participation of our patients.”
“Persons of African descent have been chronically underrepresented in clinical research, have suffered significantly higher rates of certain diseases, and have exhibited different efficacy responses to medicines, as well as different adverse drug event profiles, than other ethnic groups,” said Arthur Holden, chairman and CEO of FGT. “We are confident that this partnership will create a highly valuable resource for the global genetic and clinical research community, and ultimately, for patients of African descent.”
Over five years, it is expected that 25,000 blood samples or cheek swabs will be collected.
| Abbott and Artus GmbH to develop two-hour PCR SARS test Abbott Laboratories of Abbott Park, Ill. has announced a worldwide marketing and distribution agreement with German-based Artus GmbH for a commercial test to detect a form of the coronavirus suspected of causing Severe Acute Respiratory Syndrome (SARS). Abbott will assit in FDA submission for the test. Developed by Artus in collaboration with the Bernhard-Nocht-Institute for Tropical Medicine (BNI) in Hamburg, Germany, the test was introduced by Artus in April to countries in Asia and Europe and has been provided to laboratories under the direction of the World Health Organization (WHO) and other major clinical sites conducting studies on the SARS virus. According to Abbott, initial results of different sample types (sputum, swabs, stool and tissue) from these WHO laboratories are promising. The test is based on PCR technology, which directly detects the virus in patient samples, and produces results in two hours. It is thought that the new test is sensitive enough to detect the virus in early stages of infection. Under the agreement, Artus will manufacture the test and Abbott will market and distribute it initially for use on the Roche LightCycler thermal cycler system in the U.S., Canada, Germany, the U.K. and Austria, supplementing Artus’ existing distribution agreements. The test will also be available on the Applied Biosystems ABI Prism 7000 Sequence Detection System for worldwide distribution. |
Cerner and Abbott will develop and co-market POC medication management system
Cerner Corporation of Kansas City, Mo. and Abbott Laboratories of Abbott Park, Ill., have entered into an agreement to develop and co-market a comprehensive point-of-care medication management system for infusion therapy. The system will be designed to integrate point-of-care infusion and diagnostic medical devices, bar coding, clinical information and knowledge-based decision support tools at the patient’s bedside.
According to the companies, the system will initially aim to improve patient safety in infusion therapy administration through a secure identification of the caregiver, patient, device and IV access site matched with physician orders, patient assessment parameters, patient medical history and hospital-specific best practice treatment protocols. The system will then be enhanced to go beyond electronic medication management and bar-coding systems. It will be designed to integrate several Cerner IT applications with Abbott’s point of care diagnostic devices. The Cerner applications are to include an integration tool kit, a drug information database, a computerized physician order entry system, a pharmacy information system and an electronic medical record system. These are to be integrated with Abbott’s point-of-care diagnostic devices, including the Precision Net products, and infusion therapy pumps, including GemStar, Plum A+, Omni- Flow 4000 Plus and LifeCare PCA3. The system also will have the ability to interface with the majority of hospital information systems and medical devices. Cerner and Abbott intend to further expand the system through additional functionality for more complex therapeutic applications and clinical specialties.
| Roche and Genome Institute of Singapore will co-develop SARS kit Roche Diagnostics of Indianapolis, Ind., and Genome Institute of Singapore have formed an agreement to co-develop a SARS detection kit based on Roche’s PCR technology. The research kit to detect the virus is expected to be available by the end of July. Dr. Ren Ee Chee, deputy director of Genome Institute of Singapore, who is principal investigator on this project said: “Until SARS is eradicated, our scientists will continue to work with the relevant authorities and organisations to find solutions in stamping out the disease. This research collaboration with Roche will enable us to tap the company’s strengths in diagnostic test development as well as their ability to make therapeutics and vaccines generally available to the public.” Dr. Ling Ai Ee, Singapore General Hospital’s senior consultant virologist, said: “We are excited by this collaboration and hope that it will yield promising medical solutions. By working with the research institute and patients, our team is able to bridge science and patient care. Right now, the priority is to find healthcare solutions as soon as possible.” Once the detection kit is developed, it will need to be validated to ensure reliability and sensitivity. Such test bedding is normally carried out at key laboratories. Dr. Ling added, “ Singapore General Hospital will be glad to team up with Genome Institute of Singapore and Roche to tackle SARS by participating in the test-bedding of the new detection kit.” |
Chiron acquires PowderJect for stronger position in global vaccines market
Chiron Corporation of Emeryville, Calif. has announced that Chiron UK-1 Limited, an indirect wholly-owned subsidiary of Chiron, has purchased 1,000,000 ordinary shares of PowderJect Pharmaceuticals plc at a price of 545 pence per share. The purchased shares represent 1.09 percent of the total outstanding ordinary shares of PowderJect. Chiron announced on May 19, 2003, that it had launched a recommended cash tender offer for all outstanding ordinary shares of PowderJect, a UK-based vaccines company. The boards of directors of Chiron and PowderJect have unanimously approved the transaction.
“We consider PowderJect to be an excellent strategic fit for Chiron,” said Howard Pien, chief executive officer and president of Chiron. “This deal will help us to achieve our goal of expanding our global vaccines business quickly in the United States while building on our platform for new products. In addition to allowing us to enter the U.S. business for vaccinations for flu, the combination with PowderJect will expand our manufacturing capabilities. We look forward to working with the people of PowderJect to create a stronger global player in the vaccines business. Chiron is committed to increasing its impact on human health worldwide through all three of its business units: biopharmaceuticals, vaccines and blood testing.”
The combination of Chiron and PowderJect will build on the two companies’ existing positions as the fifth and sixth largest vaccines businesses in the world, respectively, and will represent a major advance in the vaccines business strategy of both companies. Chiron and PowderJect had combined total annual revenue of over $1.5 billion for the year to March 31, 2003.
The combined companies will be the world’s second-largest provider of flu vaccines. PowderJect’s product, Fluvirin, is a leading flu vaccine in the United Kingdom and is one of only two available injectable flu vaccines in the United States. PowderJect’s strong position in the United States is complemented by Chiron’s prominent position in Europe. With its three brands — Agrippal S1, Begrivac and Fluad — Chiron is currently the second-largest producer of flu vaccines outside of the United States.
The combination of Chiron and PowderJect is also expected to enhance the combined companies’ position as a leading player in both the United States and European vaccines businesses. Furthermore, it will provide a stronger platform for new products, including Chiron’s flu cell culture vaccines, meningococcal vaccines and novel vaccines for viral infections currently in development, to be introduced in the United States.
This acquisition will increase manufacturing capability. PowderJect’s FDA-approved manufacturing facility at Liverpool in the United Kingdom is one of the largest flu vaccines manufacturing facilities in Europe.