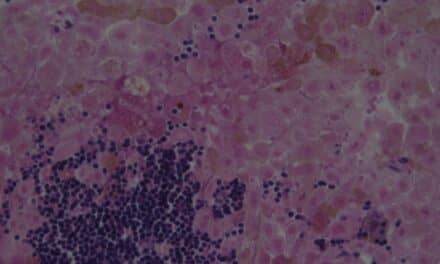
Protagen AG, Dortmund, Germany, and Gustave Roussy, Villejuif, France,will partner to utilize Protagen’s SeroTag technology to help identify biomarkers that predict and monitor immune-related adverse events (irAEs) in cancer patients treated with checkpoint inhibitors.
Checkpoint inhibitors offer potential for the treatment of many cancer indications, including melanoma and renal cell carcinoma. Yet, only a subset of patients respond favorably to treatment, and it is not currently possible to predict which patients will benefit from therapy. In addition, checkpoint inhibitors can trigger severe irAEs, which has in the past led FDA to halt clinical trials.
Through their collaboration, Protagen and Gustave Roussy will utilize Protagen’s proprietary immune system profiling platform, SeroTag, to monitor patients, detect irAEs, and ultimately conduct comprehensive risk profiling for those undergoing cancer immunotherapy.
The project is part of the ongoing Gustave Roussy immunotherapy program, which aims to develop immunotherapy access and best practices.
“Although immunotherapies like checkpoint inhibitors have shown great promise for treating those suffering from cancer, response rates for these therapies are still low, often around 10% to 20%,” says Aurelien Marabelle, MD, PhD, clinical director of the Gustave Roussy immunotherapy program. “In addition, our efforts to improve therapeutic outcomes via the implementation of combination therapies can increase the risk of the patient developing debilitating and sometimes fatal irAEs.”
Marabelle adds, “It is therefore vital that we try to understand more about the immunological responses patients are exhibiting to cancer, both before and during therapy. Utilizing Protagen’s SeroTag platform will enable us to ask these questions, and we very much look forward to this collaboration.”
“Our unique SeroTag technology has already demonstrated its ability to stratify patients into homogenous disease subgroups for a number of autoimmune indications, thereby supporting the development of novel therapies,” says Peter Schulz-Knappe, PhD, chief scientific officer at Protagen. “Due to the strong link between immunooncology and autoimmune disease, we believe that applying our technology and approach to the immunooncology field will result in improved patient selection for novel immunotherapies and support the risk profiling of patients for the development of irAEs. We feel privileged that Gustave Roussy shares this view and we are excited about our collaboration.”