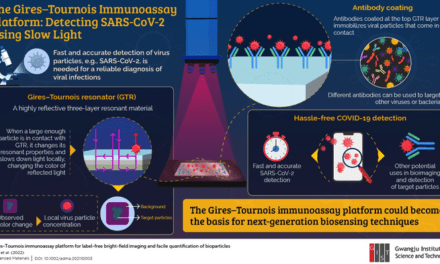
Diazyme Laboratories, Poway, Calif, has obtained CE-IVD marking for its SARS-CoV-2 neutralizing antibody test. The Diazyme test is a high-throughput chemiluminescent immunoassay with excellent correlation to a fluorescence-based neutralization assay, which has been used to evaluate the adaptive immune response in vaccine studies. Diazyme also announced plans to work with US FDA for EUA application.
“Unlike the widely available total SARS-CoV-2 IgG antibody tests, Diazyme’s neutralizing antibody test measures the subset of neutralizing antibodies that block the interaction between the receptor binding domain (RBD) of the viral spike glycoprotein and the human ACE2 cell surface receptor,” says Chong Yuan, PhD, managing director of Diazyme Laboratories. “The Diazyme neutralizing antibody test gives the scientific community and health authorities a valuable tool to evaluate the level of effective antibodies generated after infection or vaccination, and to assess adaptive immunity in studies going forward. Diazyme serology tests meet real world expectations from healthcare professionals and have been independently evaluated in several published studies.”
For more information, visit Diazyme.
Featured image: Diazyme’s SARS-CoV-2 neutralizing antibody test is run on the fully automated DZ-Lite 3000 Plus chemiluminescence analyzer, pictured. (Courtesy: Diazyme)