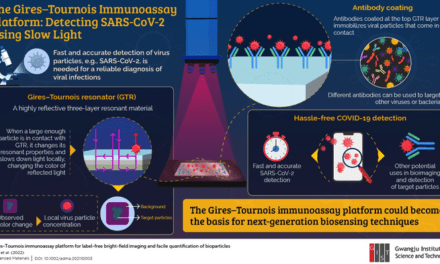
On January 11, 2022, the U.S. Food and Drug Administration (FDA) issued a safety communication warning test users, caregivers, and healthcare personnel to stop using the LuSys Laboratories COVID-19 Antigen Test (Nasal/Saliva) and the LuSys Laboratories COVID-19 IgG/IgM Antibody Test.
The performance of these tests has not been adequately established and FDA believes there is likely a high risk of false results when using these tests. Neither test has been authorized, cleared, or approved by FDA for distribution or use in the United States.
The LuSys Laboratories COVID-19 Antigen Tests (Nasal/Saliva) and COVID-19 IgG/IgM Antibody Tests may also be sold under the company names Luscient Diagnostics or Vivera Pharmaceuticals, or with the trade name EagleDx. The FDA believes that these tests were distributed for use in laboratories or for at-home testing.
The FDA is encouraging labs to review the safety communication in detail and share it widely with their networks.