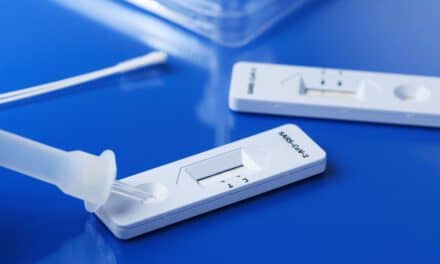
FDA has authorized the first antibody test intended for use at point-of-care settings such as doctor’s offices, hospitals, pharmacies, urgent care centers, and emergency rooms rather than having to be sent to a central lab. Carolina Liquid Chemistries Corp. (CLC), Greensboro, NC, is the official distributor of this test, which is called the Fastep COVID-19 IgG/IgM Rapid Test Device by Assure Tech of Hangzhou, China. This test is now authorized for use with fingerstick whole blood specimens only at the point-of-care, that is, in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation.
FDA Commissioner Stephen Hahn commented that authorizing point-of-care serology tests will enable “more timely and convenient results” for individuals who want to understand if they have previously been infected with the virus that causes covid-19.
After months of guidance indicating that tests intended for the point of care must be used in labs, the agency found this test met the standards to also be used with fingerstick whole blood specimens at point-of-care settings operating under CLIA Certificate of Waiver.
This covid-19 IgG/IgM Rapid Test Device was first authorized in July to detect antibodies against the virus in venous whole blood, serum, and plasma to help identify individuals with antibodies SARS-CoV-2 indicating recent or prior covid-19 infection. On September 23rd, this 15-minute lateral flow test received authorization for point-of-care use using finger-stick blood samples. External controls are also available for this product.
For more information, visit Carolina Liquid Chemistries.