A new corona test developed at the University Hospital Bonn can analyze a large number of swabs simultaneously using sequencing technology and has a similarly high sensitivity as the common qPCR test. The new method offers potential for systematic testing in daycare centers, schools or companies. The results of the study on the new corona test have been published in the Nature Biotechnology.
“Our corona test LAMP-Seq can detect about 100 times lower amounts of virus than current rapid antigen tests and is almost as sensitive and specific as the common qPCR test” describes Jonathan Schmid-Burgk, PhD, from the Institute of Clinical Chemistry and Clinical Pharmacology of the UKB the characteristics of the test procedure, which was developed interdisciplinary with other researchers at the UKB.
“Added to this is the high scalability of the test. By using sequencing machines, thousands of samples can be analyzed simultaneously,” said Schmid-Burgk, who was appointed to the University of Bonn from the Broad Institute of MIT and Harvard in early 2020. The LAMP-Seq method detects not only corona infections with the original SARS-CoV-2 virus, but also the novel variants of concern alpha to delta.
Members of the ImmunoSensation2 cluster of excellence, the Institute of Hygiene and Public Health, Life&Brain GmbH and Bundeswehr Central Hospital Koblenz were among those involved in the project.
LAMP-Seq Methodology
For the LAMP-Seq test, the Bonn scientists have adapted the already established LAMP method (Loop-mediated Isothermal Amplification – propagation of the viral genome at a constant temperature) and made it compatible with sequencing machines used for biomedical research. As a result, many samples can be analyzed simultaneously in a high-throughput procedure. Before thousands of samples are analyzed together in a sequencing run, each individual sample is linked to a molecular barcode. This barcode ensures that each sample can be assigned without doubt, even after thousands of samples have been pooled. “Retesting of the entire pool in case of a positive test result is therefore no longer necessary” says Kerstin Ludwig PhD, Emmy-Noether group leader at the Institute of Human Genetics. This technology significantly reduces the cost per test in comparison to the qPCR test and makes the LAMP-Seq procedure a scalable corona mass test.
“With its high throughput and sensitivity, the LAMP-Seq test can make a significant contribution to the screening of undetected infections. Especially in schools or companies, where many people regularly meet, the corona test is ideal to systematically and preventively monitor the occurrence of infections” describes Ludwig, the co-developer of the test procedure, the possible application scenarios of the LAMP-Seq test.
Prof. Wolfgang Holzgreve, Dr med. Dr.h.c.mult, MBA, FACOG, FRCOG, medical director and CEO of the UKB, explains the benefits of the new test for corona surveillance as follows: “In order to effectively contain a pandemic, infected people must be found before they infect others. To achieve this goal, we need mass screenings with the highest sensitivity that can give us a detailed picture of existing chains of infection. This is exactly what the corona test LAMP-Seq developed at UKB is suited for.”
Even smaller models of the sequencing machines used are capable of analyzing around 10,000 samples in a single run (duration: around ten to twelve hours). This virtually eliminates laboratory capacity as a limiting factor in testing.
In several large studies, including school and employee testing, with a total of around 20,000 tests, the Bonn scientists have extensively tested, optimized and successfully validated the entire upstream and downstream logistics, from sample collection by throat swabs to fully digital feedback of the test results.
Looking Ahead
While the Bonn scientists have currently focused their method entirely on SARS-CoV-2 testing, LAMP-Seq can also be used in the future for differential diagnostics in testing for other viruses such as influenza A and can also be quickly adapted to other viruses.
The scientists are currently working on CE certification in order to make the LAMP-Seq test available internationally in the near future. Until this approval is obtained, the technically and scientifically fully validated LAMP-Seq method will continue to be used for pilot testing.
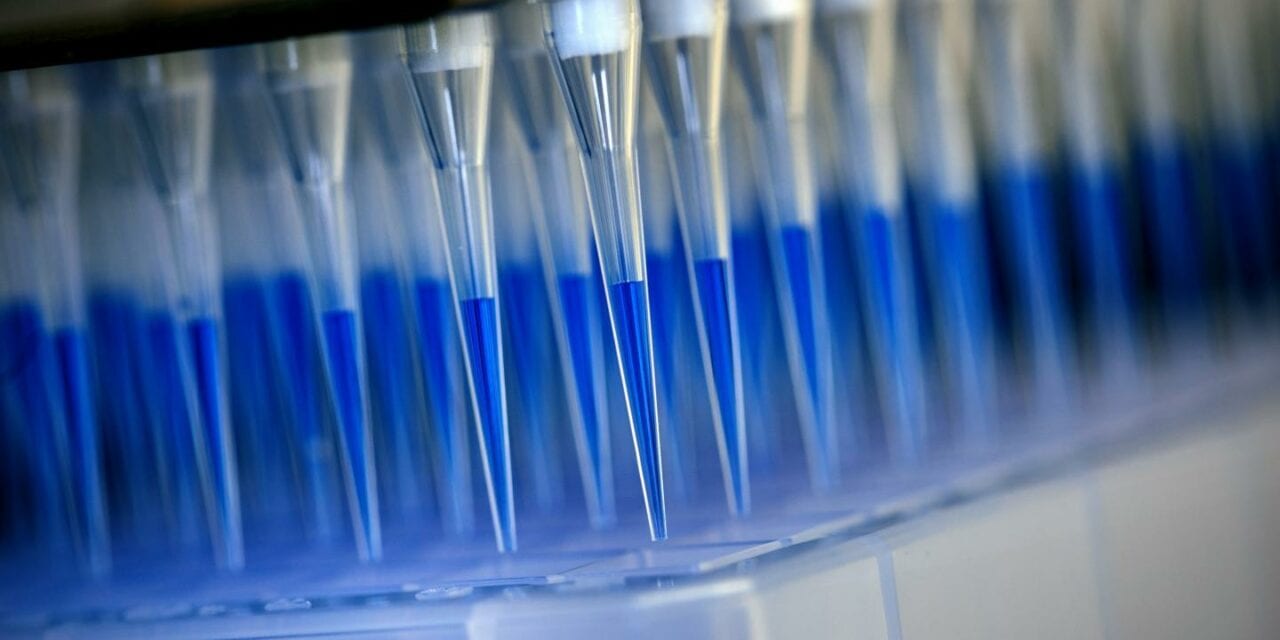
Featured Image: Blue-stained swab material from corona tests is prepared for analysis in a sequencing device with the help of a laboratory robot. Photo: Felix Heyder / University Hospital Bonn