CellCarta Secures Exclusive Global Rights to Deploy Aspyre Lung Assay in Lung Cancer Clinical Trials
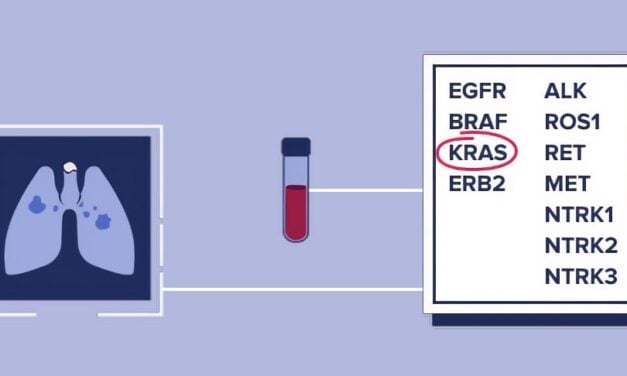
CellCarta has secured exclusive global rights to deploy Biofidelity’s Aspyre Lung assay in lung cancer clinical trials, streamlining genomic testing for trial sponsors and reducing reliance on multiple vendors.
Read More