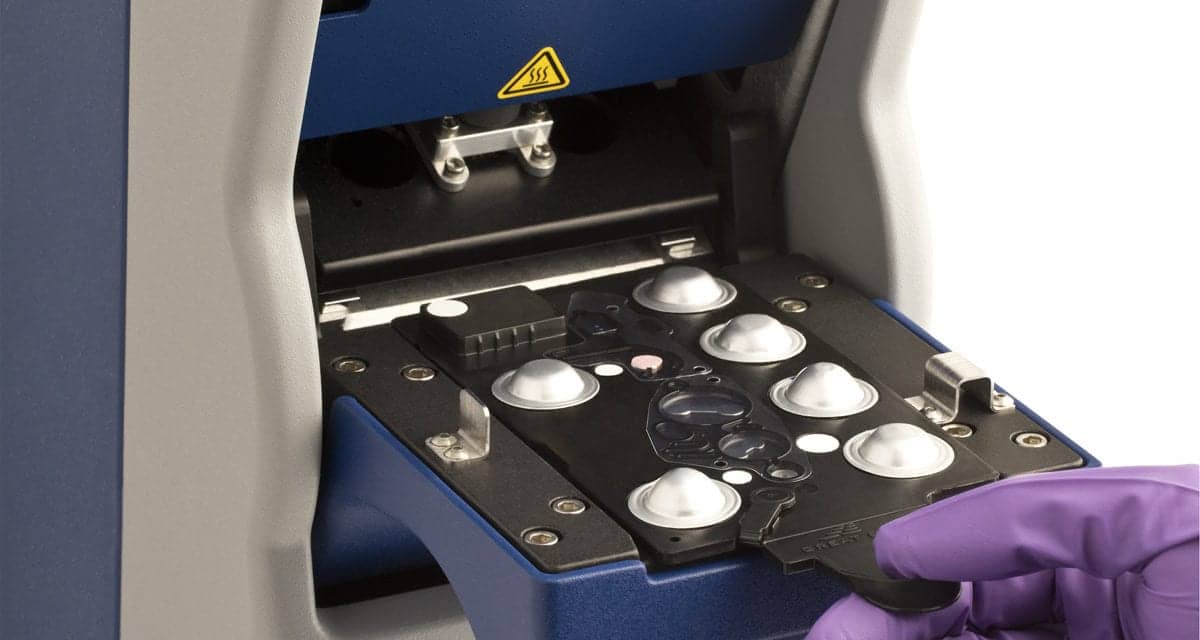
After the successful completion of a clinical trial that met all its clinical objectives, Great Basin Scientific Inc, Salt Lake City, has submitted its Staph ID/R blood culture panel to FDA for 510(k) clearance.
The company’s first multiplex panel, the Staph ID/R blood culture panel is a sample-to-result, automated, molecular multiplex panel that detects Staphylococcus aureus along with the mecA gene, a major drug-resistance marker that confers resistance to methicillin and creates the superbug MRSA.
In addition to Staphylococcus aureus, the panel also detects Staphylococcus lugdunensis and Staphylococcus epidermidis. The remaining Staph species are detected as a group and identified as “Staphylococcus present.” When compared to traditional blood culture methods, results of this panel are expected to allow for a faster diagnosis and therefore a more timely appropriate therapy, which is associated with improved patient outcomes, lower treatment costs, and shorter length of hospital stay for affected patents.
“The successful trial completion and submission to FDA of our Staph ID/R panel marks a significant milestone for Great Basin, and a critical advancement for our customers and hundreds of thousands of patients in the United States who are affected by Staph bloodstream infections—including the deadly superbug MRSA—every year,” says Ryan Ashton, cofounder and chief executive of Great Basin Scientific.
“I am proud of the determination of the Great Basin team, which continues to execute at the highest level, delivering on the promise to provide current and future customers with a robust menu of easy-to-use and cost-effective tests and panels,” Ashton says. “This panel will be higher value than our lowplex tests and increases our potential revenue per customer by offering higher revenue per test than either our C. diff test or our group B Strep test.”
Once cleared and commercially available, hospital and lab technicians can run the new multiplex panel on the same analyzer used to perform Great Basin’s commercially available lowplex tests for Clostridium difficile and group B Streptococcus.
For more information, visit Great Basin Scientific.