The molecular diagnostic services (MolDx) program of Medicare administrative contractor Palmetto GBA has issued a final local coverage determination for the MyPath melanoma test from Myriad Genetics Inc, Salt Lake City.
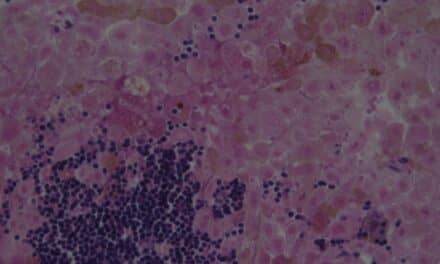
The MyPath melanoma test helps physicians provide a definitive diagnosis when a suspicious skin lesion is equivocal based upon histopathology. The test is clinically validated for use as an adjunct to histopathology when the distinction between a benign nevus and a malignant melanoma cannot be made confidently by histopathology alone. The test measures the expression of 23 genes and accurately distinguishes melanoma from benign nevi.
“We are excited that Palmetto has reviewed the extensive clinical dossier for the MyPath melanoma test and decided to cover the test for Medicare patients,” says Vicki Fish, vice president for dermatology at Myriad Genetics. “We look forward to making the MyPath melanoma test accessible to more patients so that they can obtain a definitive diagnosis and receive appropriate treatment and better health outcomes.”
Melanoma is one of the fastest growing cancers in the United States and can strike people of all ages, races, and skin types. More than one million skin biopsies are performed annually, and approximately 15% of patients have an equivocal skin lesion.
“There is strong demand among physicians for an objective genetic test to be used as an adjunct to historical approaches for diagnosis,” says Loren Clarke, MD, a board-certified dermatopathologist and medical director for dermatology at Myriad Genetics. “The MyPath melanoma test has enormous potential to help patients because an appropriate treatment plan begins with an accurate and definitive diagnosis.”
For further information, visit Myriad Genetics.