The US Department of Health and Human Services (HHS) has issued a new guidance document that specifies additional data that must be reported to HHS by laboratories along with novel coronavirus disease-2019 (covid-19) test results.
The guidance standardizes reporting to ensure that public health officials have access to comprehensive and near real-time data to inform decisionmaking in their response to covid-19. As the country begins to reopen, access to clear and accurate data is essential to communities and leadership for making decisions critical to a phased reopening.
“HHS and the entire Trump administration are deeply concerned that covid-19 is having a disproportionate impact on certain demographics, including racial minorities and older Americans,” says HHS Secretary Alex Azar. “High-quality data is at the core of any effective public health response, and standardized, comprehensive reporting of testing information will give our public health experts better data to guide decisions at all levels throughout the crisis.”
The new reporting requirements will provide information needed to better monitor disease incidence and trends by initiating epidemiologic case investigations, assisting with contact tracing, assessing availability and use of testing resources, and anticipating potential supply-chain issues.
“The requirement to include demographic data like race, ethnicity, age, and sex will enable us to ensure that all groups have equitable access to testing and allow us to accurately determine the burden of infection on vulnerable groups,” says Adm. Brett P. Giroir, MD, assistant secretary for health. “With these data we will be able to improve decisionmaking and better prevent or mitigate further illnesses among Americans.”
Laboratory data serves not only as important information to support decisionmaking related to the public health emergency, but also as a critical piece to better understand the effects of the pandemic on socially vulnerable populations. Laboratory testing data, in conjunction with case reports and other data, also provide vital guidance for mitigation and control activities.
For more information, visit the Department of Health and Human Services.
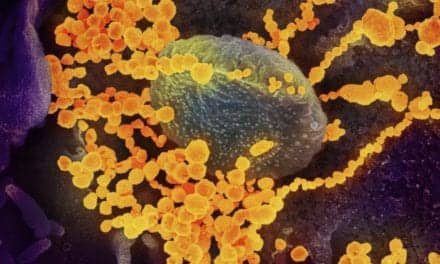
Featured image: Colorized scanning electron micrograph of an apoptotic cell (purple) heavily infected with SARS-COV-2 virus particles (yellow), isolated from a patient sample. Image captured at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID.