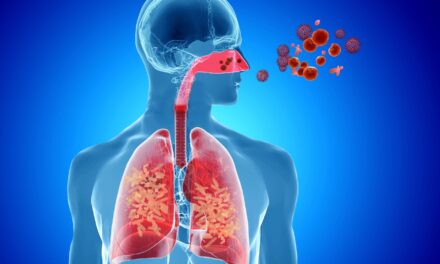
FDA Grants CLIA Waiver for Rapid Test That Differentiates Bacterial and Non-Bacterial Respiratory Infections
The FDA has granted a CLIA waiver for FebriDx, enabling its use in over 300,000 U.S. point-of-care settings and potentially reaching 80 million patients annually, according to PHASE Scientific.