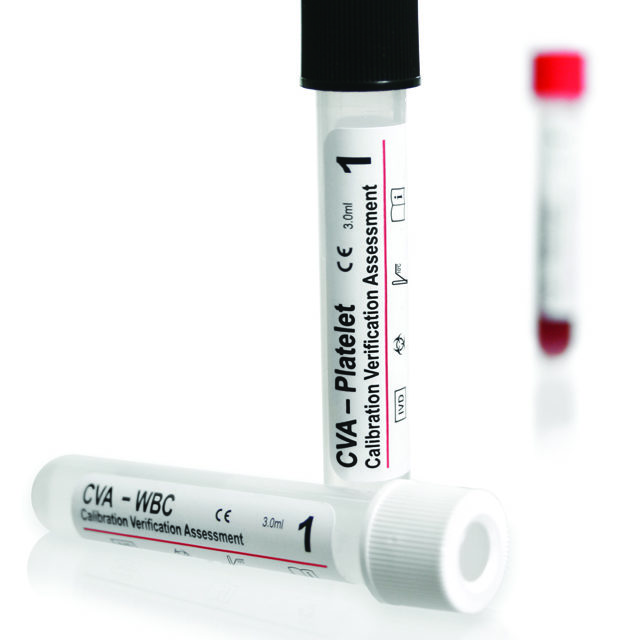
The calibration verification assessment (CVA) linearity control kit from Streck, Omaha, Neb, is now assayed for the ABX Micros 60 instrument from Horiba, Irvine, Calif. CVA is used to determine the reportable range and linear performance of three-part and five-part hematology analyzers. Each kit has customized ranges appropriate for each instrument model and provides instrument-specific assay values for the hemoglobin, platelet, red blood cell, and white blood cell (WBC) parameters. WBC and platelet parameters have extended ranges providing values up to 410,000 and 3.5 million for instruments with extended linearities. Data can be submitted to Streck for a comprehensive linearity report at no additional charge. Reports include a graph that clearly displays the instrument’s linearity and accuracy, a peer group comparison, and an interpretive results section that can demonstrate compliance with laboratory regulations. For more information, visit AACC booth 2552 or Streck.
Instrument Added to Linearity Control Assay