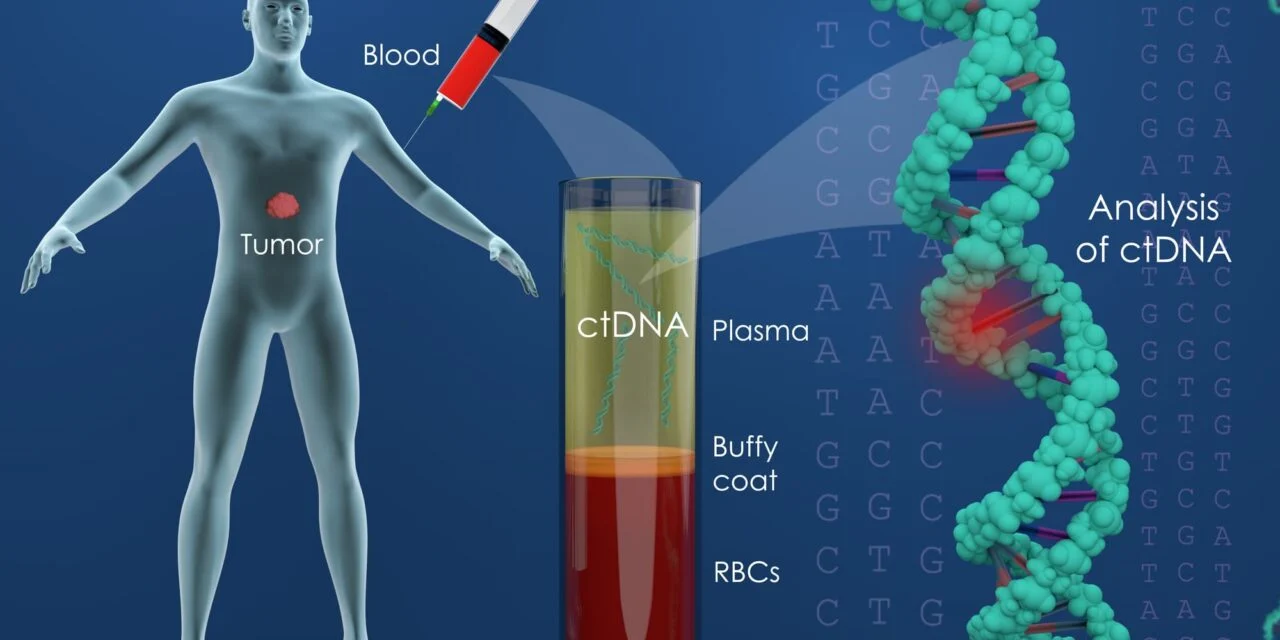
A novel enrichment method produces virtually pure circulating tumor DNA from blood samples, showing strong sensitivity and specificity across colorectal and lung cancer patients.
A new liquid biopsy method has demonstrated the ability to detect more than 95% of stage I and II cancers in a blinded validation cohort, according to proof-of-concept data released by Volition, a multinational epigenetics company.
The technology, called Capture-Seq, works by enriching and purifying plasma samples to produce virtually pure circulating tumor DNA (ctDNA) for sequencing analysis — addressing one of the core challenges in liquid biopsy, where the vast majority of circulating DNA in blood plasma originates from healthy cells rather than tumor cells.
Blinded Validation Results
The blinded validation cohort included 81 subjects — 59 colorectal and lung cancer patients and 22 healthy controls. Across all cancer stages, the test achieved an overall sensitivity of 93% and a specificity of 95%. Stage-specific detection rates were as follows:
Stage I: 94% (17/18)
Stage II: 96% (26/27)
Stage III: 100% (2/2)
Stage IV: 91% (10/11)
“We are now releasing the data from a blinded validation cohort of 81 subjects and are extremely encouraged by the results, particularly in early-stage cancer detection,” says Jake Micallef, chief scientific officer, Volition, in a release.
Micallef also noted that Volition believes it is the first liquid biopsy company to focus on circulating cell-free nucleoproteins, and has filed a number of patents to protect the technology.
Clinical Significance for Multi-Cancer Early Detection
From a clinical standpoint, the results carry implications for the multi-cancer early detection (MCED) space, where there remains a significant unmet need for reliable, non-invasive screening tools.
“The early-stage cancer detection of 95% of stage I and II cancers is particularly noteworthy,” says Andrew Retter, medical consultant, Volition, in a release. “…If validated in larger cohorts, CTCF Capture-Seq could contribute to multi-cancer early detection, fulfilling a significant unmet clinical need.”
Retter also noted that the technology may have applications beyond initial detection, including minimal residual disease detection and treatment monitoring, either alone or in combination with other diagnostic approaches.
The Technology Behind Capture-Seq
At the core of the method is a two-step process that enriches and purifies ctDNA from blood plasma, producing sequencing data sets that are greater than 99% pure cancer-derived DNA. While the method captures a subset of available ctDNA rather than the full complement present in a sample, the result is a virtually pure cancer DNA sequence set — enabling a novel class of potentially thousands of liquid biopsy sequence biomarkers, according to the company.
The underlying manuscript has been submitted for publication.
Licensing Interest and Commercial Outlook
Volition reports active discussions with several large liquid biopsy and diagnostic companies to accelerate development and commercialization of the platform.
“This scientific breakthrough has generated a lot of interest with potential licensing partners, and we are excited to share this early-stage detection data,” says Gael Forterre, chief commercial officer, Volition, in a release.
Forterre added that the company sees potential applications for the technology in both human and veterinary markets, and that it fits within Volition’s existing Nu.Q product portfolio. The company estimates the total addressable market for the human MCED application at approximately $23 billion on an annualized basis, with the MRD market representing an additional opportunity of over $13 billion.
ID 211668166 © Meletios Verras | Dreamstime.com
Related Reading:
Volition Presents New Liquid Biopsy Method for Early-Stage Cancer
Nucleosome Liquid Biopsy Shows Promise for Lung Cancer Risk Stratification
Interim Results of VolitionRx Clinical Trial Show Promise for Colorectal Cancer Screening
New ctDNA Assay Aims to Detect Molecular Residual Disease Earlier in Solid Tumors