Researchers have developed a lensless microscopy that captures cellular-level, 3D details in living tissue, looking inward to image micron-scale targets like cells and blood vessels inside the body, even through the skin.
The Bio-FlatScope is the latest iteration of lensless microscopy being developed at Rice University. Jacob Robinson, is an electrical and computer engineer at Rice’s George R. Brown School of Engineering who led the recent effort to test Bio-FlatScope in living creatures.
The research team’s FlatCam, a lensless microscopy device that channels light through a mask and directly onto a camera sensor for imaging, aimed primarily outward at the world at large. The raw images looked like static, but a custom algorithm used the data they contained to reconstruct what the camera saw.
The new lensless microscopy device looks inward to image micron-scale targets like cells and blood vessels inside the body, even through the skin. Bio-FlatScope captures images that no lensed camera can see — showing, for example, dynamic changes in the fluorescent-tagged neurons in running mice.
One advantage over other microscopes is that light captured by Bio-FlatScope can be refocused after the fact to reveal 3D details. And without lenses, the scope’s field of view is the size of the sensor (at close range to the target) or wider, without distortion.
A small, low-cost Bio-FlatScope could eventually look for signs of cancer or sepsis or become a valuable tool for endoscopy, says Robinson, who teamed with colleagues at Rice’s Neuroengineering Initiative on the project.
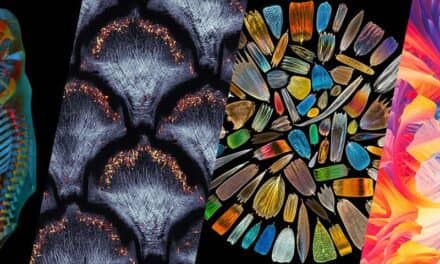
The team’s proof-of-concept study also imaged plants, hydra and even, to a limited degree, a human. Their results appear in Nature Biomedical Engineering.
The mechanism combines a sophisticated phase mask to generate patterns of light that fall directly onto the chip, according to the researchers. The mask in the original FlatCam looks something like a bar code and limits the amount of light that passes through to the camera sensor. But it doesn’t work well for biological samples.
The Bio-FlatScope phase mask looks more like the random map of a natural landscape, with no straight lines.
“Being random allows the mask to be pretty diverse in gathering light from all directions,” says postdoctoral researcher Vivek Boominathan, one of four lead authors on the study. “And then we take the random input, which is called Perlin noise, and do some processing to get these high-contrast contours.”
At the sensor, light that comes through the mask appears as a point spread function, a pair of blurry blobs that seems useless but is actually key to acquiring details about objects below the diffraction limit that are too small for many microscopes to see. The blobs’ size, shape and distance from each other indicate how far the subject is from the focal plane. Software reinterprets the data into an image that can be refocused at will.
Researchers first captured cellular structures in a lily of the valley, then calcium activity in tiny, jellyfishlike hydra. They moved on to monitoring a running rodent, attaching the Bio-FlatScope to a rodent’s skull and setting it down on a wheel. The data showed fluorescent-tagged neurons in a region of the animal’s brain, connecting activity in the motor cortex with motion and resolving blood vessels as small as 10 microns in diameter.
In collaboration with Rebecca Richards-Kortum and research scientist Jennifer Carns from Rice Bioengineering, the team identified vascular imaging as a potential clinical application of the Bio-FlatScope. Graduate student and co-lead author Jimin Wu offered her lower lip to see if light passing through to the camera could deliver structural details of the blood vessels within.
“It was kind of an engineering challenge because it’s difficult to position the Bio-FlatScope at the correct position and keep it there,” Wu says. “But it showed us it could be a good tool for seeing signs of sepsis, because pre-sepsis changes the density of the vasculature. Cancer also alters the morphology of the microvasculature.”
Long term, the team sees potential for a camera that could curve around its subject, like brain tissue, “so it could match the morphology of what you’re looking at,” Robinson says. “Or maybe you could fold it up, stick it in place and have it unfold and deploy. You could also do really interesting things by bending it for a fisheye effect, or you could curve it inward and have very high light-collection efficiency.”
Co-lead authors of the study include Rice alumnus Jesse Adams and Applied Physics graduate student Dong Yan. Co-authors are Rice graduate students Sibo Gao and Soonyoung Kim; postdoctoral alumnus Alex Rodriguez; Caleb Kemere, an associate professor of electrical and computer engineering and of bioengineering; and Ashok Veeraraghavan, a professor of electrical and computer engineering.
Richards-Kortum is the Malcolm Gillis University Professor, a professor of bioengineering and electrical and computer engineering and director of the Rice360 Institute for Global Health Technology. Robinson is an associate professor of electrical and computer engineering and of bioengineering, and he, Kemere and Veeraraghavan are core members of the Rice Neuroengineering Initiative.
The Defense Advanced Research Projects Agency (N66001-17-C-4012), the National Institutes of Health (RF1NS110501) and the National Science Foundation (1730574) supported the research.
Featured image: A comparison of images from Rice University’s Bio-FlatScope, left, and original FlatScope, right, show how much detail the new device captures in images of cells in a lily of the valley. Bio-FlatScope’s random phase mask proved better at imaging curved structures. Photo: Robinson Lab/Rice University