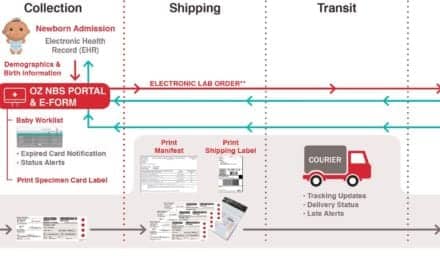
Audit MicroControls Inc, Eatonton, Ga, has expanded its line of calibration verification and linearity controls with the Linearity FD bilirubin controls for Roche systems. The set of controls is designed to simulate human serum samples for determining linearity, calibration verification, and verification of the reportable range for quantitative assays performed on Roche clinical chemistry analyzers. The Linearity FD set is a ready-to-use lyophilized product consisting of five levels that demonstrate a linear relationship to each other when assayed for direct bilirubin and total bilirubin. The product has an open-vial stability of 2 days when stored at 2–8°C. Along with the controls, the company offers Auditor QC, a free, online, real-time, data-reduction program. The package provides Levey-Jennings charts and linearity graphs, as well as peer group analysis for daily quality control product users. Report results are produced immediately and stored on the Auditor QC Web site for future reference. For more information, visit Audit MicroControls.
Linearity Set of Bilirubin Controls for Roche Clinical Chemistry Systems