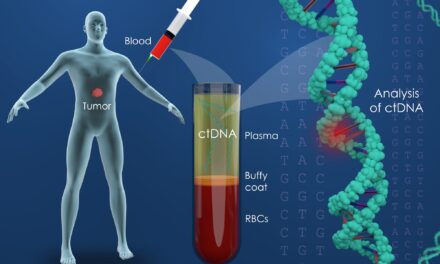
Summary: Mainz Biomed N.V. announced positive topline results from a pooled clinical study, validating the efficacy of their next-generation colorectal cancer (CRC) screening tool integrating proprietary mRNA biomarkers, with a sensitivity for CRC of 92% and specificity of 90%, setting a new standard in the field.
Takeaways:
- Advanced Screening Accuracy: Mainz Biomed’s new CRC screening tool achieves a sensitivity of 92% for colorectal cancer and 82% for advanced adenoma, surpassing existing commercial products and highlighting its potential for early detection and prevention.
- Global Validation: The pooled study included patients from Europe and the U.S., enhancing the credibility of the results by evaluating a diverse set of patient cohorts, and providing robustness to the tool’s efficacy across different populations.
- Disruptive Potential: With its high sensitivity and specificity, Mainz Biomed’s CRC screening tool, pending FDA approval, could disrupt the at-home diagnostic screening market, potentially becoming the new gold standard for accurate and reliable early detection of colorectal cancer, offering a significant improvement over existing methods.
Mainz Biomed N.V., a molecular genetics diagnostic company specializing in the early detection of cancer, announced topline results from a pooled clinical study which included new patients and subjects from Mainz Biomed’s ColoFuture (Europe) and eAArly DETECT (U.S.) clinical trials evaluating the potential to integrate its portfolio of proprietary novel gene expression (mRNA) biomarkers into a next generation version of the company’s colorectal cancer (CRC) screening tool. The topline results confirm the positive efficacy results previously reported with a sensitivity for colorectal cancer of 92% with a specificity of 90% and a sensitivity for advanced adenoma of 82%, which is best-in-class and compare favorably to existing commercial products.
“We conducted this pooled study to fine-tune the minimal biomarker set to be used to optimize sensitivity and specificity of the results in Mainz Biomed’s next generation CRC screening tool. The new data read-out demonstrates that our next generation product candidate for early-stage CRC detection utilizing mRNA biomarkers, a FIT test and a proprietary AI algorithm has consistently delivered high sensitivity and specificity for both advanced adenomas and colorectal cancer,” says Guido Baechler, chief executive officer of Mainz Biomed. “This strong performance is particularly noteworthy as the pooling a significantly higher number of patients, namely two entirely separate patient cohorts from two continents as well as previously unexamined patient samples. Hence these results represent a critical milestone on our path to launching our FDA PMA pivotal study ReconAAsense, which is planned to recruit up to 15,000 patients.”
For Further Reading: Mainz Biomed to Present Results of Colorectal Cancer Screening Study
The pooled results include 690 evaluable subjects across 21 sites in the U.S. and 9 sites in Europe. The two cohorts included patients (US cohort aged 45 and older and European cohort aged 40 and older) that provided a stool sample before undergoing a colonoscopy to either screen for colorectal cancer (average risk), to follow up on a positive non-invasive test, imaging or symptoms, or if a subject was already identified as having colorectal cancer but before any treatment had been administered. Following colonoscopy and any applicable histopathology, subjects were classified into groups: CRC, advanced adenoma, non-advanced adenoma, no findings, or non-colorectal cancer. Each subject outcome was compared to the results from the next generation test incorporating the novel mRNA biomarkers and FIT.
The company says it plans to publish results of this study at a major medical conference during the second quarter of 2024.
Colorectal Cancer Early Detection Is Critical
According to the Centers for Disease Control and Prevention (CDC), colorectal cancer is the second most lethal cancer in the U.S. and Europe, but also the most preventable, with early detection providing survival rates above 90%. Mainz Biomed’s proprietary portfolio of mRNA biomarkers has previously demonstrated the ability to detect CRC lesions, including advanced adenomas, a type of pre-cancerous polyp often attributed to this deadly disease .
The power to determine lesions in a pre-cancerous stage can change the entire colorectal cancer diagnostic field, by treating the patient before the polyps can progress to a cancerous stage. This is especially remarkable as recent studies reveal that blood tests, despite their perceived convenience, lack the sensitivity to detect pre-cancerous conditions effectively. In the landscape of early colon cancer diagnostics with non-invasive tests, stool-based screening methods proved to be the most precise. Subject to a positive outcome of the FDA PMA study, Mainz Biomed’s innovative next generation test has the potential to disrupt the at-home CRC diagnostic screening market by providing the most robust and accurate test and become new gold standard.