San Diego-based Quidel has received FDA emergency use authorization (EUA) to market its QuickVue SARS Antigen test, a point-of-care assay for the rapid, qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in anterior nares (NS) swab specimens directly from individuals who are suspected of covid-19 by their healthcare provider within the first 5 days of the onset of symptoms.
Quidel’s new QuickVue SARS Antigen test requires no supplemental instrumentation and is visually read. It offers excellent performance for the detection of SARS-CoV-2 (96.6% PPA versus PCR and 99.3% NPA versus PCR) in anterior nares swab samples, yielding results in just 10 minutes, thereby providing quick, reliable results to patients, their families and healthcare workers alike.
“We are proud to introduce yet another powerful diagnostic tool to aid the fight against covid-19,” says Douglas Bryant, president and CEO of Quidel Corporation. “The QuickVue rapid antigen test for coronavirus leverages our proven QuickVue visually read diagnostic platform for influenza A+B to further democratize access to affordable and highly accurate covid-19 testing across a diverse range of medical and point-of-care locations.”
“The flexibility of QuickVue for meeting the urgent testing needs of everyone from school systems to rural areas and even locations without electricity gives us the opportunity to do enormous good in communities across our nation and the world,” says Bryant. “We will scale immediately to supply the more than 30,000 QuickVue professional market customers we serve today and look forward to extending the benefits of this technology as broadly and rapidly as possible in the months and years ahead.”
Quidel plans to reach a production run rate of 600 million QuickVue tests per year by the end of 2021.
For more information, visit Quidel.
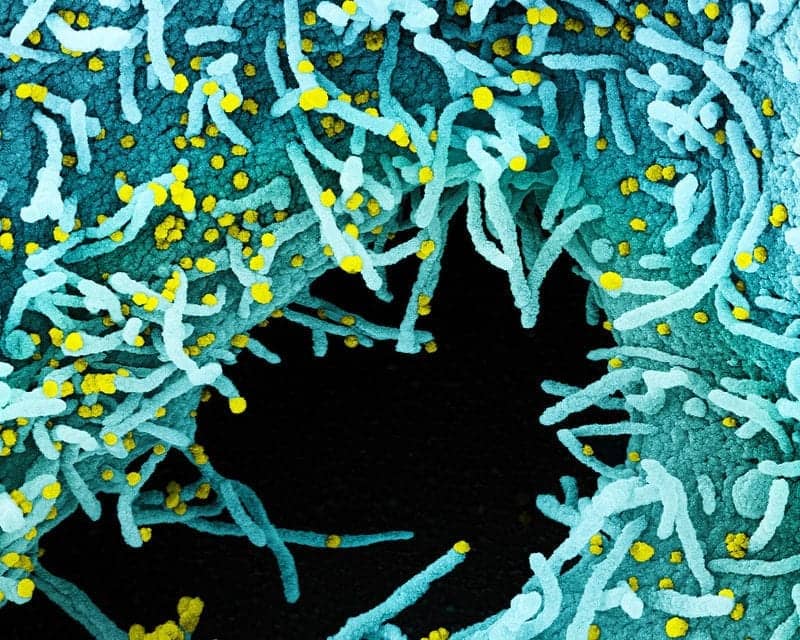
Featured image: Colorized scanning electron micrograph of a cell heavily infected with SARS-CoV-2 virus particles (yellow), isolated from a patient sample. The black area in the image is extracellular space between the cells. Image captured at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID