Makers of LIS and middleware systems offer an inside view of lab connectivity status
By Karen Appold
Clinical laboratories in the United States have long been involved in initiatives to implement fully integrated patient information systems throughout the nation’s healthcare sector. But that doesn’t mean the pace of adoption has always been steady or even.
To find out where labs stand today, CLP asked several makers of laboratory information systems (LIS) and middleware products what their recent experience has told them about the connectivity status of labs—both internally and with larger elements of the healthcare system. Their responses are as varied as their client base, but suggest that there is significant unevenness in the way that labs are addressing connectivity—and that systems are still improving in response to labs’ needs.
CONNECTED, OR NOT
One area in which labs have been especially active is in the integration of results from point-of-care devices into patient records stored in their organization’s LIS. “Point-of-care testing continues to be one of the fastest-growing hospital segments, with more and more tests moving from the lab to the bedside,” says Steven Valorz, informatics marketing manager at Alere Informatics, Charlottesville, Va.
“Today, virtually 100% of all US hospitals run glucose tests at the bedside, with other tests—such as blood gas, coagulation, and urinalysis testing—not far behind. From a connectivity standpoint,” says Valorz, “nearly 90% of all US hospitals use data management systems to transfer the testing done at the bedside to the LIS for posting to the patient’s electronic medical record.”
But what goes for point-of-care devices may not be true in other parts of labs’ operations. “Many laboratories, even in US hospitals, still have no LIS—or one that is not interfaced to the hospital’s or practice’s electronic medical records,” says Janet L. Chennault, vice president at Schuyler House, Valencia, Calif. “The largest impediment to this project is the extreme variation in interpretation of the HL7 standard.”
ADDRESSING ISSUES
Among the difficulties confronting would-be adopters of new laboratory information systems and middleware solutions is the need to have more than just a run-of-the-mill record-keeping system. Systems not designed with the specific needs of clinical labs in mind may not be suitable.
“We commonly find that electronic medical records and hospital information system vendors have designed their systems with an incomplete knowledge of the scope of laboratory work,” says Chennault. “Many systems have trouble with culture and sensitivity results.
“Other frequent problems include an inability to handle reflex tests, or any testing that generates an unexpected result set—such as discovering urine crystals,” she adds.
Given increasing laboratory workloads, simplicity of operation is also an important factor in making a new system acceptable. “The number-one need with any LIS or middleware system is ease of use. It needs to be user-friendly because at the end of the day, the lab technologist doesn’t want to have to troubleshoot an issue with the software,” says Jean-Philippe Guyon, US clinical marketing manager for
bioMérieux, Durham, NC.
“Part of that ease-of-use factor is addressing how technologists want to interface with systems moving forward,” says Guyon. “The old paradigm—where you had to go to a specific computer station—is outdated. The interface needs to be Web-based, accessible via mobile devices, and available to multiple touch points in the lab.”
PRODUCT OVERVIEWS
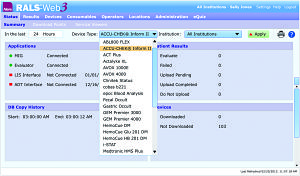
User interface of the web-based RALS-Web3 point-of-care device management system by Alere Informatics.
With such varied needs in the laboratory marketplace, makers of LIS and middleware solutions have a lot of high-value targets to be addressed. Some systems may be especially suited for some specific purposes, but not others, so labs seeking to implement new solutions may need to familiarize themselves with a number of competing products. Below is a small sampling of just a few LIS and middleware products available in today’s marketplace.
[reference float=”right”]LIS and Middleware Tech Guides
The CLP Tech Guides for January 2014 offer additional information about products from the following LIS and middleware manufacturers. Visit the guides here.
LIS
- Apex Healthware
- CompuGroup Medical Inc
- GenoLogics Life Sciences Software
- HEX Laboratory Systems
- MEDCOM Information Systems Inc
- NovoPath Inc
- Orchard Software
- Psyche Systems Corp
- SCC Soft Computer
- Schuyler House
- Sunquest Information Systems
Middleware
- Apex Healthware
- Beckman Coulter
- Data Innovations
- GenoLogics Life Sciences Software
- NovoPath Inc
- Orchard Software
- Psyche Systems Corp
- Sysmex America Inc
- Telcor Inc
[/reference]
Alere Informatics offers a number of point-of-care device management systems, including RALS-Freedom, AegisPOC, and RALS-Web3. Each of the company’s vendor-independent solutions is totally Web-based; collectively, they are used by more than 2,000 hospitals throughout the United States.
RALS systems are interfaced directly to meters and devices used at the point of care, eliminating device-specific data managers. RALS manages, configures, and communicates directly with each device, improving point-of-care program efficiency and productivity.
The company’s unified approach means that operators need to deal with just one program, data manager, reporting source, and format. “RALS systems also enable point-of-care coordinators to manage all operators that use meters and devices connected to the system,” says Valorz. “Operators can be certified for their competency using training modules within the RALS system.”
RALS systems feature direct-to-device interfaces for a number of hospital point-of-care glucose meters, including the Abbott Precision Xceed Pro, Abbott FreeStyle Precision Pro, Nova Biomedical StatStrip, and Roche Accu-Chek Inform II. RALS systems also offer connectivity with many testing devices for cardiac markers, blood gases, coagulation, and urinalysis.
Myla, by bioMérieux, is an innovative middleware solution that has been intelligently designed by and for laboratory microbiologists. ”Myla plays a central role in the pathway from sample collection to patient decision, helping labs achieve the most out of their existing instrumentation and staff,” says Guyon. “By changing the way lab microbiology information is managed, Myla ensures that laboratory professionals can recapture valuable time through improved connectivity, workflow, and information management.”
Myla provides an actionable, comprehensive picture of all testing activity in the lab, allowing for a faster, freer flow of information. This has a significant impact on patient management and ultimately, patient care. “By streamlining the flow of information in the lab, Myla helps to free up time for technicians to get full use of their expertise and skills,” says Guyon.
[reference id=”36852″]Case Study: Integrating LIS and Middleware Solutions[/reference]
In the next version of Myla, bioMérieux will introduce new features, such as built-in intelligence that will bring potentially contaminated samples to the immediate attention of the lab technologist.
SchuyLab—the first graphical LIS in the market—is offered by Schuyler House. “We continue to focus on providing an intuitive user experience,” says Chennault. “This approach minimizes training time and encourages an efficient workflow process.

SchuyLab’s wide array of optional features allows it to easily manage the work of larger laboratories. “Our satisfied users include 5,000-bed hospitals overseas and reference labs performing thousands of accessions per day in the United States,” Chennault says.
SchuyLab has more than 200 live HL7 interfaces with different electronic medical record and electronic health record systems. The company created approximately 100 different sets of interface specifications in order to communicate with these different systems.
Telcor Inc, of Lincoln, Neb, offers QML, a middleware product designed specifically to manage an organization’s point-of-care testing program. QML is a device-vendor-neutral product that connects any device used outside the clinical laboratory, where samples are primarily identified by a patient’s identification (medical record or account number) rather than a laboratory accession number. “QML currently connects to more than 95 different device types, which is many times more than any other system in this market space,” says Becky Clarke, executive vice president at Telcor.
QML can be installed in many different configurations to meet the requirements of the organization’s policies. It can reside on virtual or physical hardware with one or multiple servers, depending on volume and IT requirements.
The system’s functionality includes interfaces (admissions, discharges, and transfers; orders; device, LIS, and electronic medical record results) that run unattended, nonstop. The system also features an extensive set of result, device, and operator management functions. System reports for statistics and metrics are all accessible through Citrix or Internet Explorer.
“QML offers unlimited electronic connectivity to reduce errors and omissions of manual charting, plus tracking and documentation of operator certification requirements, and management through exception processing,” says Clarke. “It is unique in that every customer runs the same code base, which includes hundreds of configuration options to make the system function for each customer’s specific needs.”
For information about more LIS and middleware solutions available for clinical laboratories, visit the online Tech Guides in the January issue of CLP, available here.
Karen Appold is a contributing writer for CLP. For further information, contact chief editor Steve Halasey via [email protected].