The newly identified aptamers match antibody-level binding strength and could support the development of lower-cost, point-of-care blood tests for neurodegeneration.
Researchers in Japan have identified the first DNA aptamers capable of binding neurofilament light chain (NfL), a blood-based protein biomarker linked to neurodegeneration, offering a potential new avenue for developing more accessible and affordable diagnostics for Alzheimer’s disease (AD).
The study, led by associate professor Kaori Tsukakoshi from the Department of Chemistry at the Tokyo University of Science and distinguished professor Kazunori Ikebukuro from the Department of Biotechnology at the Tokyo University of Agriculture and Technology, was published in Volume 796 of Biochemical and Biophysical Research Communications. The research was conducted as part of a joint UK–Japan project supported by AMED-SICORP.
NfL is a structural protein found in large nerve fibers. When neurons are damaged, NfL is released first into the cerebrospinal fluid and then into the bloodstream, making it a key emerging biomarker for monitoring neurodegeneration that begins years before clinical symptoms of AD appear.
“We have reported the world’s first DNA aptamer that binds to NfL, which is released into the blood in response to neurodegeneration in various neurodegenerative diseases. The developed aptamer has a binding affinity comparable to commercially available antibodies, and is expected to have a variety of applications in the future, such as diagnosing the progression of AD,” says Tsukakoshi, in a release.
How the Aptamers Were Identified
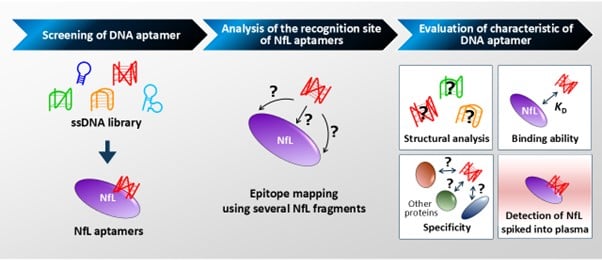
The aptamers were generated using Systematic Evolution of Ligands by Exponential Enrichment (SELEX), a process in which large libraries of random single-stranded DNA sequences are screened across multiple cycles to isolate the strongest and most selective binders. The team conducted seven rounds of selection, recovering and amplifying DNA strands that attached to NfL while removing sequences that bound to unrelated proteins. This process produced 86 candidate aptamers, which were further narrowed to 30 sequences that successfully recognized the full-length NfL protein.
Two candidates—named MN711 and MN734—emerged as the most promising. MN711 demonstrated a binding dissociation constant of 11 nM, while MN734 achieved 8.1 nM, levels the researchers say are comparable to the antibodies used in currently available commercial NfL assays. Both aptamers showed high specificity for NfL, binding selectively over other AD-related biomarkers including amyloid β and phosphorylated tau.
The aptamers were found to recognize a specific region of the NfL protein—amino acid residues 281–338—a segment known to be present in NfL fragments detected in human blood plasma. Critically, the binding activity of both aptamers was preserved when the protein was introduced into human plasma, supporting their potential utility in a blood-based diagnostic format.
Advantages Over Antibody-Based Methods
Current blood tests for NfL depend on immunoassay platforms that use antibody pairs to achieve high analytical sensitivity. While these platforms perform well, antibodies can be costly to produce, prone to batch-to-batch variability, and difficult to adapt for use in newer biosensor formats.
DNA aptamers offer several practical advantages. As chemically synthesized molecules, they can be manufactured with greater consistency and at lower cost than antibodies. They can also be modified with functional chemical groups that allow them to attach directly to electrode surfaces used in electrochemical biosensors—a property that makes them well-suited for integration into compact, point-of-care testing platforms.
“A key advantage of DNA aptamers is their compatibility with chemical modification. Aptamers can be synthesized with terminal functional groups that enable straightforward immobilization onto metallic or carbon-based electrode surfaces commonly used in electrochemical biosensors. This property allows aptamers such as MN711 and MN734 to be integrated into compact sensing platforms designed for point-of-care testing,” says Tsukakoshi, in a release.
The research was co-authored by second-year master’s student Miyu Matsumoto, also from Tokyo University of Science.
Photo credit: Tokyo University of Science
Related Reading:
Siemens Healthineers Expands Brain Health Research Portfolio with New Blood-Based Tau Assays
Lucent Diagnostics Partners to Bring Blood-Based Alzheimer’s Testing to Community Settings
Circular Genomics Secures Exclusive Rights to Blood Test Technology for Alzheimer’s Disease
Finger Prick Blood Test for Alzheimer’s Diagnosis Enters Large-Scale Trial
Study Links Fat Transport Molecules to Alzheimer’s Risk Based on Genetics