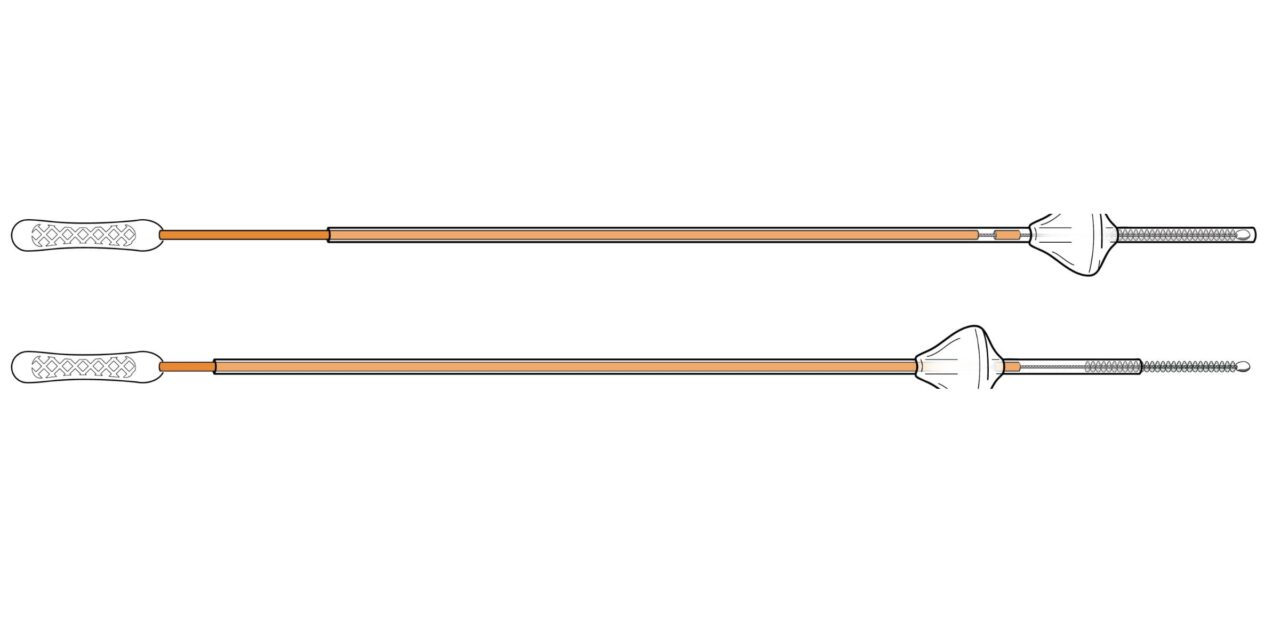
The Utepreva Endometrial Sampler utilizes three mechanisms to improve tissue yield for cytologic and molecular analysis.
Utepreva LLC announced the launch of its Utepreva Endometrial Sampler, a single-use device cleared by the Food and Drug Administration (FDA) to support the early diagnostic evaluation of endometrial cancer.
Uterine cancer is the most common gynecologic malignancy in the US, with rising incidence and mortality rates. According to the American Cancer Society, an estimated 68,270 new cases will be diagnosed this year. Clinical outcomes are highly dependent on timing, as survival rates reach 95% when the disease is identified at an early stage but fall below 20% in advanced disease.
The device is designed to modernize office-based sampling by addressing limitations of single-mechanism tools that can compromise tissue yield and delay diagnosis. It integrates three mechanisms of action: gentle tissue disruption, controlled suction, and optimized absorption via a sponge tip.
“Our mission is to save lives by expanding access to minimally invasive uterine evaluation and aiding clinicians in the detection of pre-cancer, when treatment is less invasive, and survival outcomes are significantly improved,” says Jeanetta Stega, CEO of Utepreva LLC, in a release.
Performance and Design
Preclinical and design verification testing conducted by Medical Murray evaluated the device against a commercially available endometrial sampler using a model of simulated tissue. Under controlled conditions, the Utepreva device captured a greater volume of simulated tissue and showed more uniform disruption across the sampling surface.
The collected material supports cytologic, histopathologic, and molecular cancer diagnostics, allowing for both cellular evaluation and advanced biomarker assessment from a single sample. The device features a slim-profile wand and an integrated cervical guard to prevent over-insertion. The in-office procedure is completed in approximately 20 seconds without the need for dilation, sedation, or operating room resources.
“By dramatically improving tissue capture in a simple office procedure, Utepreva aims to make a similar impact on pre-cancer detection to that of Pap smears and mammography,” says Stega in a release.
Clinical Application
The device is currently recommended for symptomatic patients, including those with abnormal or postmenopausal bleeding, and women at elevated risk due to age, obesity, or family history of endometrial, colon, or ovarian cancer. For these patients, timely and accurate tissue sampling is critical to early diagnostic evaluation.
The sampler will be available to healthcare providers in October 2026. Utepreva will present the technology at the American College of Obstetricians and Gynecologists Annual Clinical and Scientific Meeting in May 2026.
Photo caption: Utepreva wand
Photo credit: Utepreva