Advanced diagnostics mean earlier and better therapy for women
By Steve Halasey
In April of this year, the Society for Women’s Health Research (SWHR) launched a year-long public awareness campaign on the importance of molecular diagnostics for protecting and improving the health of women.
The campaign was announced during the society’s annual gala event, with a film highlighting the powerful impact of molecular diagnostics on women’s health—including coronary artery disease, breast cancer, ovarian cancer, colorectal cancer, sexually transmitted diseases, and mental health. The film is expected to be the cornerstone of a national multimedia campaign that will include print, TV, and radio public service announcements that highlight the benefits of molecular diagnostics for everyday women.
Why now? “There seems to be so much going on—more than there ever has been in the past—and a lot of it has to do with women’s health,” says Phyllis Greenberger, SWHR president and CEO. “Since we often use diagnostics to focus in on sex differences in health and disease—and specifically to understand how a lot of conditions affect women—using genomics to understand these issues is very consistent with our mission in women’s health.”
Advanced diagnostics are helping to uncover the underlying mechanisms of disease and expanding the scope of the understanding of women’s health issues. Based on this understanding, clinicians are able to customize testing options, which in turn promotes early detection, less-invasive diagnosis, and therapies that take into account the key genetic differences between men and women.
“We already see the impact of molecular diagnostics on women’s health in a wide range of diseases and conditions,” says Greenberger, “and as they continue guiding targeted and individualized treatment, healthcare providers will increasingly be able to provide the right care to the right patient at the right time.”
To get an idea of how these advances are developing, CLP took a look at some of the recent developments in cancer research, where molecular diagnostics in particular are showing special promise.
BREAST CANCER
According to the National Cancer Institute, roughly one in eight women will be diagnosed with breast cancer in their lifetime. Women with a mutation in either the BRCA 1 or BRCA 2 gene have risks of up to 87% for developing breast cancer and up to 40% for developing ovarian cancer by age 70. Mutations in BRCA genes are responsible for the majority of hereditary breast and ovarian cancers.
Previously, breast cancer detection was possible only after cancer had developed. But the development of molecular diagnostics promises to enable clinicians to effectively predict and therefore prevent hereditary breast cancer. Testing for BRCA gene mutations can now be accomplished via a simple blood test or oral rinse, enabling physicians to act to reduce cancer risk, delay the onset of cancer, and detect cancer at an earlier and more treatable stage or before cancer has a chance to develop. Moreover, women who do not test positive for the BRCA mutation can avoid unnecessary treatments and medical interventions.
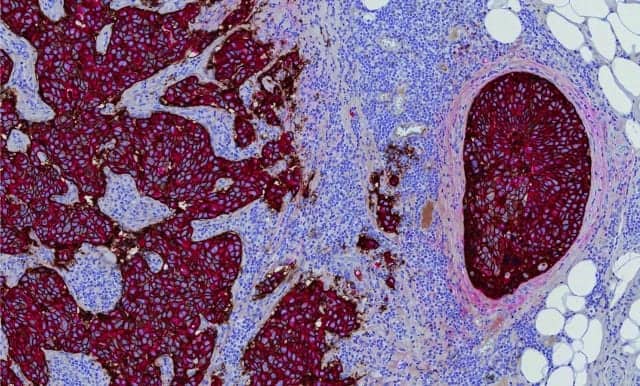
Even casual observers of research in the field of breast cancer are aware that molecular diagnostics are making significant strides toward advancing detection, therapy selection, and monitoring of breast cancer. But it has taken time—and some significant technological advances—for the field to gather speed.
“It’s been just a little more than a decade since we saw the introduction of the first molecular diagnostic tests in breast cancer. At the time, the technologies required to provide these tests were so complex that they could only be handled by a few centralized esoteric testing labs,” says Brad Gray, CEO of NanoString Technologies, Seattle. “Over the past decade, technology has become more robust and simplified, allowing the same laboratories that perform basic pathology workup of breast cancer to now also provide advanced molecular diagnostics.”
Advances in the field have been achieved by both clinical testing labs and IVD manufacturers, each contributing to the design and development of clinically useful tests (Table 1). Among the molecular and other achievements of the past few years:
- Agendia, Irvine, Calif, has developed the Symphony genomic breast cancer profile, consisting of four assays directed at specific clinical questions: MammaPrint to determine the risk of recurrence; BluePrint to help physicians select therapies; TargetPrint to confirm receptor status for ER, PR, and HER2 to help guide hormonal therapy; and TheraPrint to provide gene expression for 56 genes with correlations to therapy response in metastatic disease.
- San Diego-based bioTheranostics Inc is among the laboratory leaders in developing molecular diagnostic tests for early-stage breast cancer. A subsidiary of French IVD giant bioMérieux, the company operates a CLIA-certified, CAP-accredited diagnostic laboratory to perform its proprietary Breast Cancer Index. The index is a molecular test that predicts early (0 to 5 years) and late (5 to 10 years) distant recurrence among patients with estrogen receptor-positive breast cancer, and is also able to predict patient benefits from extended endocrine therapy. The ability of the Breast Cancer Index to predict which patients will benefit from extended endocrine treatment is significant in the context of safety concerns and side effects. “Extended tamoxifen treatment is known to double the risk of endometrial cancer, ischemic heart disease, and pulmonary embolism over a 5-year period,” says Richard Ding, CEO of bioTheranostics. “And aromatase inhibitors such as letrozole and exemestane are known to cause cumulative bone loss that can lead to osteoporosis and fractures. Women on endocrine treatments also often suffer from substantial tolerability challenges, such as hot flashes, vaginal discharge, joint stiffness, and gastrointestinal issues.”
- Janssen Diagnostics, Raritan, NJ, a business unit within Johnson & Johnson’s Janssen Pharmaceuticals subsidiary, has developed the CellSearch circulating tumor cell kit, which measures the number of circulating tumor cells (CTCs) in the patient’s blood. CTCs are associated with decreased progression-free survival and decreased overall survival in patients treated for metastatic breast cancer. The technology is also applicable to monitoring therapy response in metastatic colorectal and prostate cancer.1,2
- Myriad Genetics Inc, Salt Lake City, developed the BRACAnalysis early detection test for sequencing of the BRCA 1 and BRCA 2 genes.
- NanoString’s Prosigna breast cancer prognostic gene signature assay is a CE-marked and FDA-cleared in vitro diagnostic assay that is performed on the company’s nCounter Dx analysis system using formalin-fixed, paraffin-embedded breast tumor tissue previously diagnosed as invasive breast carcinoma. The qualitative assay uses gene-expression data, weighted together with clinical variables, to generate a risk category and numerical score expressing a patient’s risk of distant recurrence of disease.
- Siemens Healthcare Diagnostics, Tarrytown, NY, has developed a serum HER 2/neu test for quantitative determination of the HER 2/neu protein in the follow-up and monitoring of patients with metastatic breast cancer. Monitoring serum HER 2/neu levels enables the determination of patient response to therapy and cancer progression.
[reference id=”38768″]Sidebar 1[/reference]
- Ventana Medical Systems Inc, Tucson, Ariz, has developed a comprehensive suite of immunohistochemistry (IHC) and in situ hybridization (ISH) diagnostic solutions for determining a patient’s ER, PR, and HER 2 expression status for hormone therapy eligibility.
Along the way to such advances, however, laboratories and IVD manufacturers have also had to contend with significant competitive issues—including the ownership of intellectual properties involving genetic sequences. At least some of these issues were resolved with the June 2013 Supreme Court ruling in the case of Association for Molecular Pathology v Myriad Genetics, in which the court held that naturally occurring genetic sequences are not patentable. The ruling essentially overthrew some of the key patents previously issued to Myriad Genetics, which had become the basis for the company’s proprietary BRCA 1 and 2 breast cancer testing services.
“This particular ruling was akin to declaring BRCA 1 and 2 testing generic,” says Gray. “Sequencing these two genes is a relatively straightforward process that can be duplicated in many labs equipped with sequencing equipment. Therefore, many labs are now competing with fairly similar tests.
“In contrast, molecular diagnostics based on gene expression are complex tests involving patented algorithms. Each test is differentiated in multiple ways,” says Gray. “For instance, NanoString’s Prosigna assay has been shown to be more sensitive in identifying high-risk patients, and it is the only test available as an in vitro diagnostic kit that can be run locally at pathology labs all over the world.”
With greater freedom to operate without infringing others’ patents, IVD manufacturers and clinical labs are able to identify and go after tough diagnostic problems considered to be clinically important. “A particular issue for breast cancer patients is late recurrence—that is, recurrence of cancer more than 5 years after diagnosis,” says Gray. “If a physician could identify patients at risk for recurrence after 5 years, those patients could be treated with endocrine therapy longer, potentially preventing the recurrence. Newer second-generation breast cancer prognostic assays, such as Prosigna, provide this risk information.”
In July, the New York State Department of Health granted approval for the Breast Cancer Index by bioTheranostics to be used for patients from New York State. Statistics show that the majority (about two-thirds) of breast cancer patients are estrogen receptor positive, and the risk of late distant recurrence remains a substantial concern, with more than 50% of recurrences occurring after 5 years in this patient group. The Breast Cancer Index has been studied extensively, with clinical research data covering more than 2,200 patients and results of pivotal studies appearing in several journals during 2013.3–5
According to NanoString’s Gray, even more promising technologies are on the horizon. “There are other possibilities that have been demonstrated in clinical studies,” says Gray, “including identifying women who are at such low risk as to not require radiation therapy, and others at high risk who are likely to respond to certain types of chemotherapy and not others.
“Newer technology platforms are addressing a fundamental challenge in cancer—the need for more biological information from too little tumor tissue,” Gray adds. “We’ve made significant progress detecting cancer earlier than before, but this means that tumors are small and little tissue is available for analysis. We’re now starting to see the cusp of what new technologies can do with limited tissue samples, and the amount of information that can be generated to inform treatment decisions.”
CERVICAL CANCER
According to the American Cancer Society, between 1955 and 1992 the US death rate from cervical cancer declined by almost 70%, largely as a result of the increased use of Pap screening. Such improvements continued with the 1996 introduction of the ThinPrep Pap test by Hologic, Bedford, Mass, with the US rate of invasive cervical cancer falling 28% since the test became available. The ThinPrep Pap test and imaging system are FDA approved or cleared for cervical cancer screening and testing for chlamydia, gonorrhea, and human papilloma virus (HPV).
The Pap test has traditionally been used to check the cervix for abnormal cells that can be removed even before they become cancerous. But with the discovery that nearly all cervical cancers are caused by persistent HPV infection, the target of greatest diagnostic interest has shifted (Table 2).
Now, the development of molecular diagnostics is making it possible to identify women at greatest risk for cervical cancer as a result of their infection with HPV. About a dozen forms of the HPV virus are considered high risk, with the overexpression of p16 or p18 correlated with roughly 70% of all cases of cervical cancer. (For more information, see the companion article, “The STD Connection“). Other advances in the field include:
- In August 2012, Quest Diagnostics, Madison, NJ, launched its cervical cancer telomerase RNA component (TERC) test as an adjunct to conventional Pap and HPV tests.
- Ventana Medical Systems Inc has developed the CINtec p16 histology biomarker to measure HPV infection and detect cervical cancer.
COLORECTAL CANCER
Colon cancer is one of the top three most common cancers among women—second among Asian/Pacific Islander women, and third among American Indian/Alaskan Native, Black, Hispanic, and White women.
According to the American Cancer Society, approximately 75% of colorectal cancers are not inherited, but are diseases of the genome—meaning that every cancerous cell has some genetic alteration. Over the past decade, research has shown the underlying basis of colorectal cancer to be an accumulation of genetic alterations. Such an understanding of the genetic roots of colorectal cancer is also providing the basis for the development of innovative methods of colorectal cancer screening (Table 3).

Senior research associate Tanya Quint loads one of the instruments used to run Exact Sciences’ Cologuard test at the company’s headquarters in Madison, Wis. Photo: Business Wire.
Leading the way in this field is Exact Sciences Corp, Madison, Wis, which has developed Cologuard, a molecular diagnostic that detects gene mutations found in stool—alerting doctors to the possible presence of precancerous polyps and colon cancer with an accuracy of more than 90%. The test also identifies the presence of blood in the stool, another indicator of possible colorectal cancer. The latest American Cancer Society colorectal cancer screening guidelines include stool-based DNA testing as a recommended screening option.
In preparation for filing a premarket approval (PMA) submission with FDA, Exact Sciences conducted several extensive studies, most recently comparing the performance of the Cologuard test to colonoscopy and fecal immunochemical testing (FIT). One of the most extensive colorectal cancer screening studies ever conducted in the United States, the company’s pivotal DeeP-C trial included 90 sites and enrolled 10,000 patients.6 Key findings of the study included:
- Sensitivity of Cologuard in detecting patients with colorectal cancer was 92% versus 74% for FIT;
- Sensitivity in detecting patients with colorectal cancers in Stages I through III —those determined by the American Joint Committee on Cancer to be associated with an increased rate of being cured—was 93% for Cologuard versus 73% for FIT;
- Sensitivity for patients with advanced precancerous lesions was 42% for Cologuard versus 24% for FIT;
- Cologuard detected 69% of patients with polyps with high-grade dysplasia versus 46% for FIT;
- Cologuard achieved a specificity of 87% versus FIT specificity of 95%.
“We know that colorectal cancer can be prevented and can be highly treatable if found early through screening,” says lead author Thomas F. Imperiale, MD, of the Indiana University School of Medicine, Regenstrief Institute Inc, the IU Simon Cancer Center, and the Roudebush VA Medical Center in Indianapolis. “Sensitivity is the most important characteristic for screening tests, because the primary role of such testing is to rule out diseases such as cancer.
“In our clinical study, the data show that Cologuard, the multitarget stool-based DNA test, is highly sensitive in detecting colorectal cancer and higher-risk precancerous polyps in a large, diverse average-risk patient population—and has the potential to be an important screening tool.”
Exact Sciences submitted its PMA application last year, and received a unanimous recommendation from the molecular and clinical genetics panel of FDA’s medical devices advisory committee at the end of March. The company is now awaiting a final decision from the agency, which analysts expect may be delivered by the end of September 2014.
In addition to its role in improving the diagnosis and monitoring of metastatic breast cancer, the CellSearch circulating tumor cell kit by Janssen Diagnostics, Raritan, NJ, is also finding application in testing for colorectal cancer. Detached from their original tumor source, CTCs use the bloodstream to travel throughout the body, and sometimes establish new tumors in other sites. In patients treated for colorectal cancer, CTCs are associated with decreased progression-free survival and decreased overall survival. Ongoing monitoring of patients during the course of their therapy permits clinicians to make adjustments if CTC levels rise.
OVARIAN CANCER
Ovarian cancer is the eighth most common cancer and the fifth leading cause of cancer death among women in the United States. According to the National Cancer Institute, nearly 22,000 women in the United States will be diagnosed with ovarian cancer in 2014, and more than 14,000 women will die from the disease. Although it accounts for just 3% of all cancers in women, ovarian cancer causes more deaths than any other gynecologic cancer.7
Patient mortality rates for ovarian cancer indicate that improvements are needed in the treatment and prevention of the disease. In spite of considerable research into therapies for ovarian cancer, the disease remains difficult to diagnose and treat effectively. It is estimated that more than 1 million women are living with ovarian masses. Of those successfully diagnosed, only 33% receive the standard care recommended by the National Comprehensive Cancer Network and the American College of Obstetrics and Gynecology (ACOG), which is for women with an ovarian mass that is suspicious for cancer to be under the care of a gynecologic oncologist. Consequently, a considerable portion of ovarian cancer patients do not have their first surgery performed by the appropriate specialist.8
Several tests are available to detect ovarian cancer and to monitor patient responses to therapy, but ongoing studies suggest that even better tools are on the way (Table 4). With the right combination of tests, experts believe, it may soon be possible to identify ovarian cancer with an accuracy of around 95%. In turn, assignment of the correct specialist to perform initial surgery promises to improve quality of care and increase survival rates by as much as 30%.
A blood test to detect levels of cancer antigen 125 (CA125) is widely used for monitoring the ongoing effects of therapy, or detecting the recurrence of cancer after treatment has ended. Because the CA125 test is an immunoassay with low sensitivity and specificity for detecting early-stage cancers, the test is not suitable for use in screening. It may be used to monitor women with a family history or genetic mutation that suggests increased risk for ovarian cancer. The test is available as a commercialized product from several manufacturers.
The blood test for human epididymal protein 4 (HE4) is also used for monitoring patients for recurrence of ovarian cancer. While the HE4 test is an immunoassay that yields a quantitative result—the amount of the HE4 protein in a patient’s blood—an elevated level of the protein is not necessarily indicative of disease recurrence because other conditions can also increase HE4 levels. Serial testing using HE4 by itself or in combination with CA125 may be requested in order to increase the chances of detecting a recurrence. The HE4 assay is available as a commercialized product from several manufacturers, including Abbott Diagnostics, Lake Forest, Ill, whose version of the test is optimized to run on the company’s Architect family of immunochemistry analyzers.
Researchers are working to improve on the performance of such older tests. Vermillion Inc, Austin, Tex, an IVD developer and manufacturer with programs in women’s health and gynecologic oncology, has adopted a novel strategy for its OVA1 test for ovarian cancer. The company’s lead diagnostic, OVA1 is a blood test for presurgical assessment of ovarian tumors for malignancy, using an innovative algorithmic approach. As the first FDA-cleared, protein-based in vitro diagnostic multivariate index assay, OVA1 represents a new class of software-based diagnostics.
OVA1 measures the levels of five tumor-secreted proteins found in the blood (including CA125), and then uses a proprietary algorithm and software called OvaCalc to calculate a woman’s risk of cancer. The resulting risk score can help physicians determine whether surgery is necessary and the most effective course of treatment.
A recent study looked at the benefit of pairing OVA1 with the two most commonly used imaging methods—ultrasound and computed tomography—to evaluate the risk of ovarian malignancy. The research team evaluated a population of 1,100 patients planning surgery for ovarian masses, and found that the combination of OVA1 with imaging reduced the proportion of ovarian cancers missed with imaging alone by 85% to 90% (Table 5). The team also developed logistic regression models to explore how menopausal status, high- or low-risk imaging, and OVA1 score interact in the assessment of ovarian cancer risk.9
The researchers concluded that “serum biomarkers and imaging are a complementary set of clinical tools, and that when the [OVA1] score is further stratified by imaging risk and menopausal status, there is a better understanding of the clinical risk of ovarian malignancy.”
“The sensitivity of OVA1 is critical to the detection of ovarian malignancy, presurgical risk assessment, and determining whether a woman may benefit from consultation with a gynecologic oncologist prior to surgery,” says Donald Munroe, chief scientific officer and SVP of business development at Vermillion.
“Recent publications have reinforced that the first surgery is an important opportunity to improve ovarian cancer survival by ensuring that cancers are detected earlier and that they are operated on by the most experienced specialists,” says study co-author Fred Ueland, MD, associate professor of gynecologic oncology at the University of Kentucky’s Markey Cancer Center. “This new study advances our understanding of how OVA1 and imaging work together in the presurgical assessment of ovarian cancer risk.”
Other researchers are continuing to define diagnostic tools that can stratify patients according to genetic groupings that may influence their response to specific drugs. In the UK, the ICON7 trial examined the effects of using bevacizumab (Avastin) in combination with standard chemotherapy for women with ovarian cancer.
In conjunction with this trial, Almac Group Ltd, Craigavon, UK, developed the ALM AADx assay, a 63-gene test that can prospectively identify a subgroup of high-grade serous ovarian cancer (HGSOC) patients that have a good prognosis following standard chemotherapy. The subgroup is characterized by the absence of angiogenesis, and represents 40% of the HGSOC population.
Equally important, the trial also revealed that the good prognosis subgroup exhibited worse progression-free and overall survival when bevacizumab was made a part of their treatment.
In findings presented at the 2014 ASCO meeting, in Chicago, Almac researchers indicated that their study had found three molecular subgroups within HGSOC: one with angiogenic gene inactivation, immune gene upregulation, and better overall survival rates (termed the “immune” subgroup); and two with angiogenic gene upregulation and a correspondingly worse overall survival rate following standard of care first-line chemotherapy.
Richard Kennedy, MD, PhD, professor of medical oncology at Queen’s University Belfast, states, “This assay may help clinicians discuss the likely benefit from standard carboplatin- and paclitaxel-based chemotherapy with their patients, and could inform discussions on other potential approaches to treatment for those with a predicted worse outcome.”
Almac believes the ALM AADx assay will also work for other anti-angiogenic compounds that function in a similar manner, and hopes to bring the test to the EU market in 2015.
CONCLUSION
In women’s health, as in other fields involving genomics, researchers are just beginning to build momentum. Every day, it seems, brings announcements about newly discovered genes with potential as biomarkers or therapeutic targets.
For the healthcare community as a whole—including both government and private payors and policymakers—a major challenge will be to avoid undermining such growth with restrictive policies or inadequate payments. At the recent “21st Century Cures” roundtable hosted by the House Energy and Commerce Committee, Personalized Medicine Coalition President Edward Abrahams summed it up simply: “If innovators do not get paid, they will not develop tests.”
For more information, visit the companion article, “The STD Connection.”
Steve Halasey is chief editor of CLP.
REFERENCES
1. Hayes DF, Cristofanilli M, Budd GT, et al. Circulating tumor cells at each follow-up time point during therapy of metastatic breast cancer patients predict progression-free and overall survival. Clin Cancer Res. 2006;12(14):4218–4224.
2. Rack B, Schindlbeck C, Jückstock J, et al. Circulating tumor cells predict survival in early average-to-high risk breast cancer patients. J Natl Cancer Inst. 2014;106(5): dju066; doi: 10.1093/jnci/dju066.
3. Zhang Y, Schnabel CA, Schroeder BE, et al. Breast cancer index identifies early-stage estrogen receptor-positive breast cancer patients at risk for early- and late-distant recurrence. Clin Cancer Res. 2013;19(15):4196–4205.
4. Sgroi DC, Sestak I, Cuzick J, et al. Prediction of late distant recurrence in patients with oestrogen-receptor-positive breast cancer: a prospective comparison of the breast-cancer index (BCI) assay, 21-gene recurrence score, and IHC4 in the TransATAC study population. Lancet Oncol. 2013;14(11):1067–1076; doi: 10.1016/S1470-2045(13)70387-5.
5. Sgroi DC, Carney E, Zarrella E, et al. Prediction of late disease recurrence and extended adjuvant Letrozole benefit by the HOXB13/IL17BR biomarker. J Natl Cancer Inst. 2013;105(14):1036–1042; doi: 10.1093/jnci/djt146.
6. Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Multitarget stool DNA testing for colorectal-cancer screening. N Engl J Med. 2014;370(14):1287–1297; doi: 10.1056/NEJMoa1311194.
7. National Cancer Institute. SEER Stat Fact Sheets: Ovarian Cancer. Available at: http://seer.cancer.gov/statfacts/html/ovary.html. Accessed July 28, 2014.
8. Earle CC, Schrag D, Neville BA, et al. Effect of surgeon specialty on process of cancer care and outcomes for ovarian cancer patients. J Natl Cancer Inst. 2006; 98(3):172–180; doi: 10.1093/jnci/djj019.
9. Goodrich ST, Bristow RE, Santoso JT, et al. The effect of ovarian imaging on the clinical interpretation of a multivariate index assay. Am J Obstet Gynecol. 2014;211(1):65e1–65e11; doi: 10.1016/j.ajog.2014.02.010.