The covid-19 assay from CQuentia, Fort Worth, Texas, has received FDA emergency use authorization. The company’s CLIA labs are now offering the testing of patient samples.
CQuentia’s covid-19 testing employs qualitative multiplexing chemistries in an automated platform for the simultaneous detection and identification of SARS-CoV-2 nucleic acid in nasopharyngeal swab specimens collected from patients. CQuentia has the capacity to process more than 20,000 samples per month.
“Now more than ever the world needs accurate molecular testing to identify those people who are at risk, infected, or immune to the novel coronavirus to better understand, manage, and contain this pandemic,” says Alan Meeker, CEO of CQuentia. “Through mobilization of CQuentia’s integrated resources, including our molecular laboratory and new covid-19 test, we believe we can offer value by providing reliable results to hospital and clinic customers in as little 1 to 3 days from receipt of specimen.”
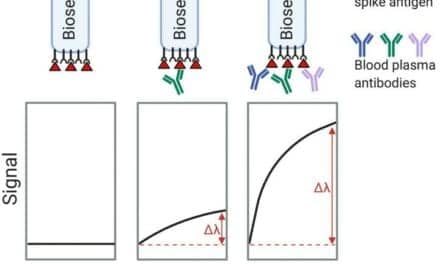
CQuentia is also validating a serological point-of-care test, based on a blood draw or fingerstick, to detect antibodies to SARS-CoV-2.
For more information, visit CQuentia.