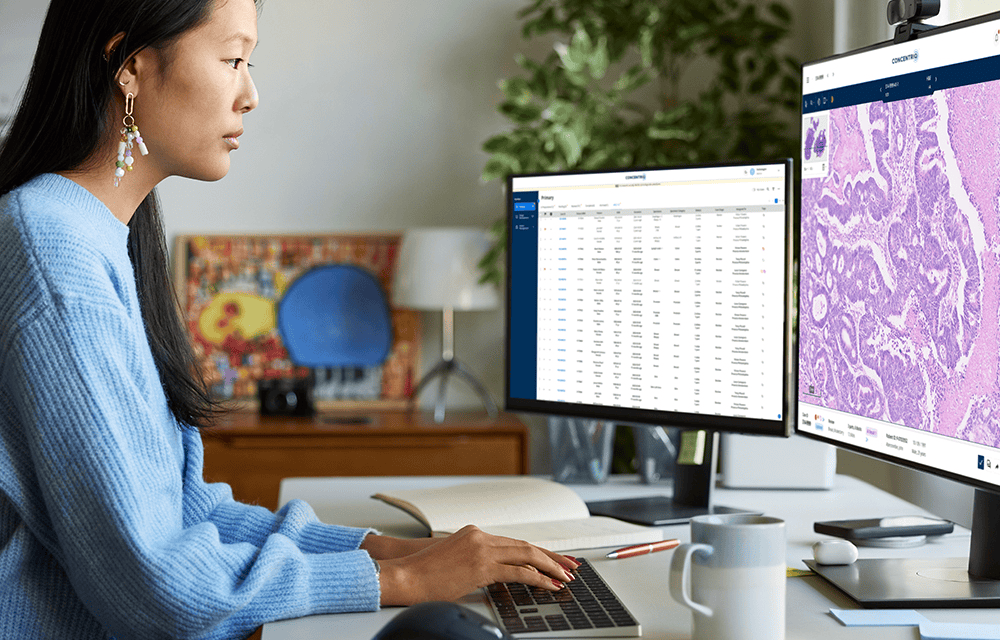
Summary: Proscia has strengthened its clinical customer base in the past year, boosted by positive outcomes from its AI-enabled workflows and further validated by a KLAS Research spotlight and a collaborative study with Quest Diagnostics.
Three key takeaways:
- Rapid Adoption and Customer Satisfaction: Proscia’s customer base growth is supported by high satisfaction rates among users, who report significant immediate benefits from using the Concentriq platform, such as improved turnaround times and enhanced research capabilities, leading to a unanimous willingness among surveyed customers to repurchase.
- Impact of AI on Pathology: The study conducted with Quest Diagnostics demonstrated the efficacy of Proscia’s AI workflows, significantly increasing the efficiency of prostate cancer diagnosis and indicating a strong potential for broader adoption of AI in pathology.
- Regulatory Advancements and Market Expansion: Proscia’s recent FDA 510(k) clearance for its Concentriq AP-Dx solution marks a significant regulatory milestone, allowing expansion into the U.S. diagnostic pathology market and enabling partnerships with major distributors like Agilent Technologies and Siemens Healthineers.
Proscia announced that it has grown its clinical customer base by more than 100% in the past year.
Proscia was also recognized by KLAS Research in a June 2024 Emerging Company Spotlight report and released findings from a study conducted with Quest Diagnostics on the impact of its AI-enabled workflows.
Proscia’s momentum comes as digital pathology experiences a new surge in global demand, according to the company. Laboratories are increasingly seeking to expand their businesses by capitalizing on the real-world data generated in the shift from microscope to images; each whole slide image contains over 1 billion pixels that can advance the next blockbuster therapies.
How Concentriq Delivers Value and Customers Trust
Early data from KLAS shows the majority of Proscia’s customers surveyed saw immediate benefits from Concentriq, and all such customers achieved outcomes within six months of deploying the enterprise pathology platform. Impacts cited include improving turnaround times, attracting more talent, and creating a real-world data archive for fueling research and development.
The KLAS report additionally reveals that these customers are impressed with both the level of support they receive and Proscia’s pathology expertise. They also awarded Proscia an ‘A+’ rating for its executive team’s involvement. Working with a trusted partner that offers a world-class software platform has led 100% of Proscia’s customers surveyed to say they would buy again.
Read KLAS Research’s Emerging Company Spotlight on Concentriq by accessing the full report here.
Quest Diagnostics study highlights Concentriq’s scalable approach to AI
Using Concentriq’s AI-enabled workflows with Ibex’s clinical-grade, AI-powered Prostate* solution, pathologists at Quest Diagnostics were able to sign out 2.9 cases per hour instead of 1.2. The offering was designed to assist pathologists in improving the detection and grading of prostate cancer by streamlining pathologists’ review of tissue samples that have been scanned at high resolution to create digital slides, or whole slide images (WSIs), which pathologists can then view.
The study included 180 randomized prostate cases from adult (21 or older) subjects. All of the pathologists who participated in the study stated they would be much more likely to adopt AI at the study’s conclusion. Quest Diagnostics, through its AmeriPath and Dermpath Diagnostics subspecialty pathology businesses, employs over 400 pathologists at its nationwide network of clinical laboratories.
“We are thrilled to help demonstrate the impact of AI-driven pathology through this study with Proscia and Ibex Medical Analytics,” says Kristie Dolan, senior vice president and general manager of Oncology & Pathology at Quest Diagnostics. “The findings provide further evidence that pathology is undergoing an unprecedented transformation, and we will continue to collaborate with world-class companies to remain at the forefront of this innovation.”
View and download the study in its entirety here.
Further reading: Proscia And Nucleai Will Broaden Access to AI Predictive Biomarkers
FDA Clears Proscia Software for U.S. Diagnostic Pathology Market
In February, Proscia received FDA 510(k) clearance for its Concentriq AP-Dx** solution for the purpose of primary diagnosis. This milestone not only opened the U.S. diagnostic pathology market to Proscia but also enables its distribution partners Agilent Technologies and Siemens Healthineers to provide its software to their U.S.-based customers, the company says.
Proscia’s 510(k) clearance further builds on its regulatory credentials. It was the first company to receive CE-IVDR certification to advance digital pathology primary diagnosis in the European Union, and has products licensed in Canada and cleared in the United Kingdom among other countries.
“More and more laboratories are putting their trust in Proscia to help them deliver faster results, provide confident diagnoses, and expand their businesses, especially now that we have FDA clearance,” says David West, CEO. “It’s incredibly rewarding to see the impact our Concentriq platform and team are delivering. This is evident from the positive feedback our customers shared with KLAS as well as Quest Diagnostics’ impressive study results.”
References:
*The Ibex suite includes solutions which are for Research Use Only (RUO) in the United States and not cleared by the FDA. Multiple solutions are CE marked (IVDD and IVDR) and registered with the UK MHRA. For more information, including indication for use and regulatory approval in other countries, contact Ibex Medical Analytics.
**Concentriq AP-Dx is cleared (K230839) for clinical use with the Hamamatsu NanoZoomer S360MD Slide scanner in the United States.
Photo: Proscia