Blood Test Using Inflammatory Biomarkers Advances for Colorectal Cancer Detection
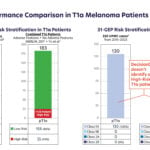
Irish researchers have secured €670,000 to clinically validate a blood-based colorectal cancer screening test using inflammatory biomarkers, aiming for launch by 2027 through a new spin-out company.