New Data Supports Multi-Omic Blood Test for Early Ovarian Cancer Detection
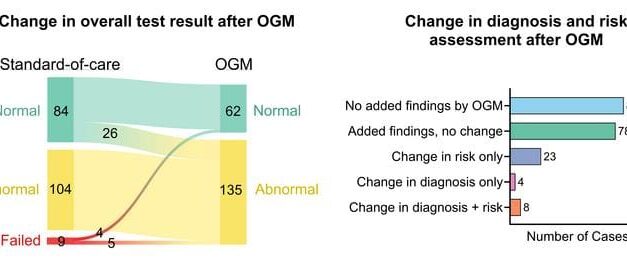
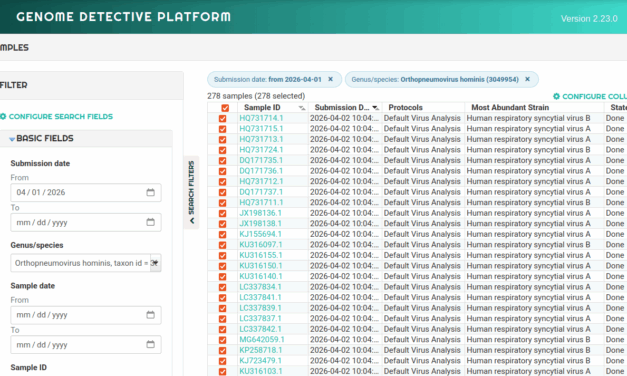
AOA Dx presented data at AACR 2026 supporting its AKRIVIS GD Test System, a multi-omic, blood-based assay for early ovarian cancer detection in symptomatic women, reporting an AUC of 0.92 in clinical controls.